CDC did something strange today: they added COVID to the Vaccines for Children program, which is a federal entitlement program that pays for vaccines for kids who don’t have other insurance. But it was not necessary to do this yet, as CDC cannot buy EUA vaccines. The COVID vaccines (as well as monoclonal antibodies, some other drugs, PCR and rapid tests) are unlicensed experimental products. EUA vaccines cannot be sold. The government buys them and your taxes pay for them.
CDC spends about 5 billion dollars a year to buy vaccines on the childhood schedule for kids on medicaid or without insurance from the manufacturers. Here is the price list.
So the vote today meant that once the COVID vaccines are sold commercially, they will be included in the Vaccines for Children program. At some time that was unspecified.
There was no clear explanation why this vote was held today, when CDC is jumping the gun. Did the CDC switch out a vote on adding COVID vaccines to the childhood schedule for a different vote at the last minute?
The CDC briefer, Jeanne Santoli, who was trying to explain why the members were voting on this, gave a very short and non-explanatory talk. I just timed her and it lasted all of 3 minutes. It was totally unlike the presentations by other briefers. Jose Romero, who is the Director of CDC’s National Center for Immunizations and Respiratory Diseases, followed her and said this is not about the childhood schedule. You can listen to them and read the slides at 6 hours 31 minutes and draw your own conclusions.
The more I think about it, the more I believe that the VFC vote was a last-minute plug-in and that CDC cancelled a vote on adding the COVID vaccines to the childhood schedule, which would cause them to be required for most American kids to attend school. The midterms are coming, after all. And people are watching.
Below is my live blog.
merylnass: It is October 19, 2022 and the CDC has scheduled a 2 day meeting of its advisory committee. However, CDC has been cagey about what they plan to change in the childhood schedule.
merylnass: There is a large vaccine lineup to be discussed:chikungunya, pneumococcal vaccines, flu vaccines, meningococcal vaccines, RSV vaccines, monkeypox vaccines and dengue vaccine.
merylnass: I had to give it a couple of extra clicks to get the show to start at https://video.ibm.com/channel/VWBXKBR8af4
merylnass: Does anyone have any memory of when the CDC or ACIP responded to comments at any time in the past? CHD encouraged comments regarding the authorization of COVID vaccines for the youngest kids–I think about 40,000 comments were sent. CDC was silent. Crickets. No response.
merylnass: Dr. Nirav Shah, the head of the Maine CDC, a smart narrative pusher, has now joined the ACIP. Dr. Kevin Ault is now gone–did he criticize the vaccines for COVID?
merylnass: Katherine Paling, Cybil Cinneas, Camille Kotton, Sarah Long, Nirav Shah,Matt Daley, Oliver Brooks, Lynn Bhata RN, Jamie Lehrer, Wilbur Chen, Keipp Talbot, Beth Bell, Ms. Veronica McNally, Dr. Sanchez, Grace Lee.
merylnass: These are the voting members. Now they go through the ex officio, nonvoting members.
merylnass: The CMS and FDA members are MIA. That is odd. Office of ID and HIV_AIDS policy is also missing—he came late. Sean O’Leary is an ex officio and he also made a comment to the last meeting, strongly in favor of covid vax for kids.
No AMA rep. Normally that would be Dr. Sandra Fryhofer. She came late. Stinchfield also made a comment last time–I’d wager the CDC asked its ex officios to weigh in and give it some support last time. Society for adolescent medicine is also missing. It is odd that so many are missing, especially the FDA member who might be needed for the discussions.
merylnass: There are several pneumococcal vaccines and clarification for which should be used when is needed. Pfizer and Merck have 2 each for adults.
merylnass: The numbers in the names refer to the number of strains aka serotypes of pneumococcal bacteria included in each vaccine. In general, the more serotypes, the more cost.
merylnass: I will again note that CDC has a 100 million dollar media center but it seems to use a $20 dollar setup for the ACIP meetings, which are chock full of delays, disconnections, and now a ten minute break. We never see the faces of the speakers and there is no livestreaming.
merylnass: The break appears to be due to difficulty connecting the speaker. These glitches may serve to cover up deliberate glitches when someone is saying something the CDC does not want the public to hear.
merylnass: Pablo Sanchez has arrived and the system is back up. Sandra Fryhofer is here too.
merylnass: No one has acknowledged any conflicts of interest
merylnass: Anyone else finding the sound keeps cutting off?
merylnass: Best I can tell, in the silence, this will be a presentation of modelling and therefore another example of how CDC hates to use real data
merylnass: Rochelle, what’s up with the $100 million media center? Suggest you try Zoom.
merylnass: Finally I refreshed the page and learned I have missed a bunch
merylnass: The guesstimate is that it costs a lot of money to save a single year of life–hundreds of thousands of dollars. Who pays the costs? Whose life is saved? If you save a 70 year old for 10 years, it might cost $4 million. Did the money come from another program or is it from moneyprinting that will debase the currency and cause inflation?
merylnass: And most of the benefits are speculative.
merylnass: How much is a life worth? In other places $100K/year or $127K/yr have been suggested. So using these models, the vaccine is too expensive given the benefits.
merylnass: Maine’s Dr. Shah is the first questioner. They can’t find slide 12. how can that be?
merylnass: Did anyone hear how long the vaccine lasts? Why do these people get so many different pneumococcal vaccines?
merylnass: No one understands this comparison, which is all modelling anyway.
merylnass: As western civilization ends, the science becomes unintelligible
merylnass: Let’s move on to something else and maybe some of the members will be able to understand that one.
merylnass: Note that PCV 23 is the OLDEST vaccine. Its efficacy was called into question at least 15 years ago, and it causes significant inflammation when given. You would think that the broadest spectrum vaccine would be the most beneficial, but all these other vaccines have been created because PCV23 is not that good. Instead of replacing it, they just keep adding more kinds.
merylnass: Now the slide show fails again. CDC is such a clown show. The Keystone Cops. 13,000 employees. A 15 billion per year budget.
merylnass: The evidence to recommendations part of the discussion is the BS part, in which all sorts of fluffy considerations are thrown together to conclude whatever CDC wants the committee to conclude. Note that her slide was based on “claims data” so you cannot check the database and make sure what she is telling you is accurate
merylnass: Invasive pneumococcal disease numbers come from CDC, but there is no reference where we can look up the data and see how it was derived.
merylnass: Now she admits they used estimates for some of the numbers.
merylnass: When PCV13 vaccine was introduced, there was no reduction in disease, and in fact a slight increase.
merylnass: Pfizer study admits the new serotypes in PCV20 only account for 3-4% of cases–not a great benefit, especially when Pfizer is telling you.
merylnass: Not sure that data gathered during the pandemic is that accurate, when people would do anything to stay out of the hospital.
merylnass: The newer vaccines “are expected” to do better than the old PCV23–wait, what? The conjugate vaccines have been available for decades and are very expensive. They still don’t know how well they work?
merylnass: Note: no PCV20 efficacy or effectiveness data
merylnass: Gee whiz, we are looking at antibodies 1 month post vax. Means nothing. And the data must suck, because they provide no numbers for us to evaluate. This is a travesty: believe what I say, as I won’t show you the data.
merylnass: Now we look at anticipated benefits, since we don’t have data to tell us the benefits. Even so, the benefits predicted are slim. They never tell us how the workgroup voted when there is questionable benefit. Then, anticipating no side effects, you might as well add this new 20 valent vaccine since of course all vaccines bring tremendous benefits and no adverse events
merylnass: Maybe the fact they were already vaccinated with older vaccines has something to do with benefit? Pfizer surveyed medical providers and they liked the vaccine, but liked it least for adults who are healthy and already had 2 different PCV vaccines.
merylnass: GIGO again. Complex assumptions that vary between models for the Pfizer and Merck vaccines, thus of limited value.
merylnass: Still quite expensive to gain years of life for people in their 70s and up–up to half a million per year to gain a bit more life–and this is based on modelling that is entirely unreliable.
merylnass: Now she fails to show how the work group voted–but I can assume they were not favorable
merylnass: The Affordable Care Act was a giveaway to vaccine mfrs. Once the vaccine is recommended by this committee, insurance is required to cover the cost 100%–no copay or deductible–by one year after the recommendation is made. This is how Obama was able to claim he was all in on prevention. Note that this allowed vaccine prices to skyrocket.
merylnass: Now they give Merck a special dispensation to weigh in!!! What did that cost Merck?
merylnass: He asks them to remember that the oldest PCV23 vaccine works, and “remains a good option.” In other words, you don’t have great data for the PCV20, and our old vaccine works (but maybe it works, unclear about that) so use us not Pfizer.
merylnass: One thing these members hate is complexity and this PCV20 vaccine addition will make a complicated schedule even more complicated.
merylnass: Dr. Brook says maybe vaccinating the kids will help the adults (and then you won’t need to vaccinate the adults, since that appears to be what happened when the Prevnar vaccines rolled out.
merylnass: Dr. Long: this is difficult stuff. The work group rarely settled on a single answer, and we were a problematic group. What they should have concluded is that there is not enough benefit to go with the new vaccine, and can revisit the issue after the vaccine is used in the population, probably by the immunocompromised, and then see how much benefit and risk it conveys.
merylnass: Dr. Shah from Maine points out the issue of having to stock so many similar vaccines. It means the clinics throw more away; it costs the clinics more; the staff make more mistakes.
merylnass: Grace Lee likes the new vaccine for the immunocompromised for 12,000 Americans. But the mfrs will not produce vaccines for such a small population and will rely on general use, once recommended.
merylnass: Helen Keipp Talbot points out that the models ignore post-hospital disability and this would make the vaccines more useful.
merylnass: Sarah Long points out that the PCV7 in kids helped adults, but the PCV13, which came out later, did not–so the PCV20 may not either. I like her.
merylnass: I suspect the cost of the PCV20, based on Prevnar costs, is likely to be $250-300 per dose.
merylnass: The last speaker implies they should approve it now cause it takes a year to get the insurers to cover it, so let’s get ahead of that
merylnass: This speaker says they should put pressure on Pharma for rational pricing. Duh. Lowered price could make cost-effectiveness better.
merylnass: Kudos to the pharmacist who represents a national organization for pointing out pharmacists generally do not have access to the medical record and cannot necessarily make good decisions and provide good advice to patients
merylnass: Current speaker said she got the PCV20 and insurance would not pay and she was charged $247 for a dose out of pocket–because it was not recommended by ACIP for a year, I guess.
merylnass: Be aware how arbitrary the decisions this committee will make will be, based on little more than speculation
merylnass: The staff are asked to provide cleaner questions for a vote. Because the members are confused by what is being asked of them. Dr. Kobayashi wants to clarify. It seems there are never too many vaccines and never too many permutations for how they can be used.
merylnass: I am avoiding all this minutiae about changes to existing recommendations. But note the vagueness of the adverse event information. You don’t learn what they are. And you get a meaningless statement that most of the reactions were mild or moderate and not severe. Well, that is true of every vaccine I know of. It conveys no information. It hides relevant information. It fills in the space where real data about adverse events should be specified with their rates, but are not.
merylnass: Ten minute break till 11:35. We are already nearly an hour behind.
merylnass: Chikungunya vaccine now. Votes on the pneumococcal vaccines later.
merylnass: No chik vaccine has ever been licensed, but I am aware of attempts to make one for probably 30 years. Which may mean there are intrinsic problems with making a vaccine for this condition.
merylnass: The US government’s DOD has long been interested in this virus, though there were no US outbreaks until 2013.
merylnass: Aedes aegypti and albopictus daytime dusk-dawn mosquitos carry it, but there can be vertical transmission, needlestick, and even airborne in labs.
merylnass: Note that we are not told how frequent the serious complications may be. Not good in pregnancy, it seems. Usually it lasts 7-10 days but there is occasional prolonged symptomatology
merylnass: It seems the studies are not very helpful. Bug repellants are the preventive.
merylnass: Valneva and Emergent BioSolutions, the crooked anthrax vaccine mfr that threw away the ingredients for 400 million doses of cOVID vaccines due to contamination is developing one vaccine–stay away from it. They also now own a cholera and a typhoid vaccine. Their typhoid vaccine made one of my patients fairly ill 2 years ago.
merylnass: There is also a Merck and an Indian candidate vaccine, both sponsored by CEPI (started by Bill Gates)
merylnass: It seems there is an interesting business plan: bring a tropical disease to the US that was never seen there before, then bring in a new vaccine for the disease. This has happened with Dengue and Dengvaxia
merylnass: They say that human challenge studies can be justified. This new unethical way of studying a vaccine is apparently now joining the ranks of acceptable clinical science.
merylnass: It looks like the Animal Rule is being invoked to get the Valneva version licensed under accelerated approval. Dr. Chen asks who is really at risk of a complicated course?
merylnass: Remember that less than 100 Americans per year get this infection, though the numbers could rise. Dr. Hills sounds stressed, as if she does not want to answer, probably due to lack of data. Though mother-fetus transmission can be devastating.
merylnass: Dr. Brooks asks how outbreaks get started–especially in the US, when the disease is so rare?
merylnass: She dodges the question of how it gets to a place where it has not previously been seen.
merylnass: Dr. Dubischar from Valneva now presents on their candidate vaccine. It is live with a genetic deletion of 60 amino acids in one protein.
merylnass: Neutralizing antibodies–human sera injected into monkeys who were then challenged with the real virus. The injected primates had no fever and most lacked live replicating virus. Some did have virus but lower titers. Valneva believes they have identified a dose that will provide sterilizing immunity, i.e., no virus growing in patients after vaccination and exposure.
merylnass: Some monkeys did grow virus, which is what is telling Valneva how large a dose to use to prevent virus in the vaccinated and exposed.
merylnass: Medium dose (3 x 10 to the 4th) viral particles is being used.
merylnass: They were allowed to skip Phase 2 by assuming that the booster dose acted as a challenge study.
merylnass: Solicited adverse events were only collected for 10 days–nice work if you can get it! Great way to avoid finding serious side effects. Vaccinees followed up to 6 months for unsolicited side effects.
merylnass: Odd that neutralizing antibodies were identical in those over and under 65 years.
merylnass: The immunogenicity seems remarkable, with antibodies in over 96% at 6 months. The side effect profiles are not good however.
merylnass: 50% had systemic AEs, some severe fevers
merylnass: 17% of recipients got joint pain vs 5% in placebo. In 3,000 subjects, one case of SIADH occurred 10 days after his shot. One developed muscle pain requiring a 5 day hospitalization. Serious AEs in 1 in 1500 recipients–but more may have been overlooked because of the short duration of active surveillance.
merylnass: 73% had any AE. Seems like you will be better off accepting a very low risk of getting chikungunya than taking a chance with this vaccine.
merylnass: Why is the ACIP looking at these vaccines before they are licensed? This makes sure there is a lot less data and less give and take because they don’t have the info that would have been presented to FDA.
merylnass: While pregnant women are the ones who would potentially get the most benefit, the company is clearly scared to death to test the vaccine in them, as it is a live vaccine and will likely cause intrauterine infection.
merylnass: 0.3% had joint pain severe enough to interfere with daily activities. Mean duration 8 days.
merylnass: Dr. Sanchez asked about the joint pains–any arthritis? And pregnancy?
merylnass: OOps. She had to admit women did get pregnant during the study and they had miscarriages and normal births and she claims it was what would be expected generally. If so, she would have presented those data as evidence of safety. The fact that she failed to present them initially suggests there is a potential problem. Dr. Sanchez asked if the fetus got infected? She says we didn’t look. That is the clincher.
merylnass: Now the speaker was instructed by a committee member to do pregnancy studies in monkeys.
merylnass: Now CDC presents again on the vax. 100 of 462 subjects were dropped due to protocol deviations–this is a high dropout rate.
merylnass: Most stopped coming back. Hmmm.
merylnass: 2% had a severe adverse event. That is VERY HIGH, especially when the placebo group only had 0.1%
merylnass: 2% of the subjects had joint pain for over 15 days. Sounds like we are normalizing prolonged side effects from vaccines now
merylnass: The data have only had a preliminary review–so why is this being presented to ACIP when it seems there is no good reason to license this, as there is almost no chikungunya disease in the US and the side effect profile is poor; furthermore, there were no data presented to convince us the disease is a serious problem.
merylnass: Except in pregnancy, for whom the vaccine is probably too dangerous.
merylnass: Someone asked about simultaneous live virus vaccines being given. I will say that in literature 20 years ago, giving lives vaccines together was said to reduce the immunity induced.
merylnass: So it was NOT RECOMMENDED to give live vaccines together. But that was when there was still a modicum of real science. I am not surprised Valneva refused to answer.
merylnass: Hills from CDC also refused to take up the question.
merylnass: Severe immunocompromise will be a contraindication to this vaccine
merylnass: What a cluster. A dangerous vaccine for which limited data are made available for what is usually a minor disease that almost does not exist in the US.
merylnass: Break till 1:30
merylnass: Next up, COVID VACCINES
merylnass: From a 2007 Senate report headed by Tom Coburn https://www.cbsnews.com/htdocs/pdf/cdc_off_center.pdf
CDC’s $106 million Thomas R. Harkin Global Communications (and Visitor) Center • CDC’s new $109.8 million Arlen Specter Headquarters and Emergency Operations Center has $10 million in furniture • CDC’s $200,000 fitness center includes $30,000 saunas and rotating light shows • CDC’s new Hawaii office announced by Hawaii Senator who oversees CDC funds
merylnass: No wonder they don’t manage to do competent reviews of vaccines; they are spending too much time in the saunas or at the Hawaii office. Remember that these amounts are 2006 dollars
merylnass: Announcement: TODAY the Novavax was approved by FDA for use as a booster in addition to an initial vaccine for adults. Despite the ACIP meeting today,the members were not asked to approve this rollout, and Rochelle Walensky has already approved it for adults. Strange omission.
merylnass: The lesson is that the fed agencies are in a RUSH to get all this investigational garbage into arms as fast as possible before the entire program explodes.
merylnass: 7 days ago the FDA granted an authorization for moderna and Pfizer boosters for kids 5 years and up.
merylnass: Ellington slipped and just said “pregnant women” and then slipped in the word people a few words later.
merylnass: The data being presented here come from a BMJ article published in 2020–when there were much more severe variants and much fewer treatments available, so it is not surprising that pregnant WOMEN had significantly worse morbidity and mortality than non-pregnant women. Pregnancy itself turns down the immune system
merylnass: While the intent of the presentation is to scare us regarding 1-6 month old babies, in fact COVID was present in only 0.5% of babies who died over the pandemic.
merylnass: This presentation uses CDC’s VSD data to assess risk of vaccination in pregnancy from the earlier vaccines. This database in earlier studies never showed a problem with the mRNA vaccines. Finally it did so re myocarditis, but has failed to find any other problems. I suspect there is a crooked method of analysis. CDC never lets us know how these numbers were derived, how they adjust them and how they analyze the raw data.
merylnass: As expected, these data claim you supposedly actually benefit slightly in terms of miscarriage from the vaccine.
merylnass: And they are assiduously studying the data and stay tuned for more happy news
merylnass: I have screenshot all the people involved in these studies for future reference so we can get them discussing these studies with their hands on a bible
merylnass: The V-safe data shows less than half the expected miscarriages in vaccinated moms. Do you believe these data? Of course not–it is impossible to have such low rates. That proves there is a problem with the study.
merylnass: Furthermore, the claim is being made that comparing vaccinated pregnant women to vaccinated pregnant women who got covid, the covid patients did not have a worse fetal outcome. How likely is that?
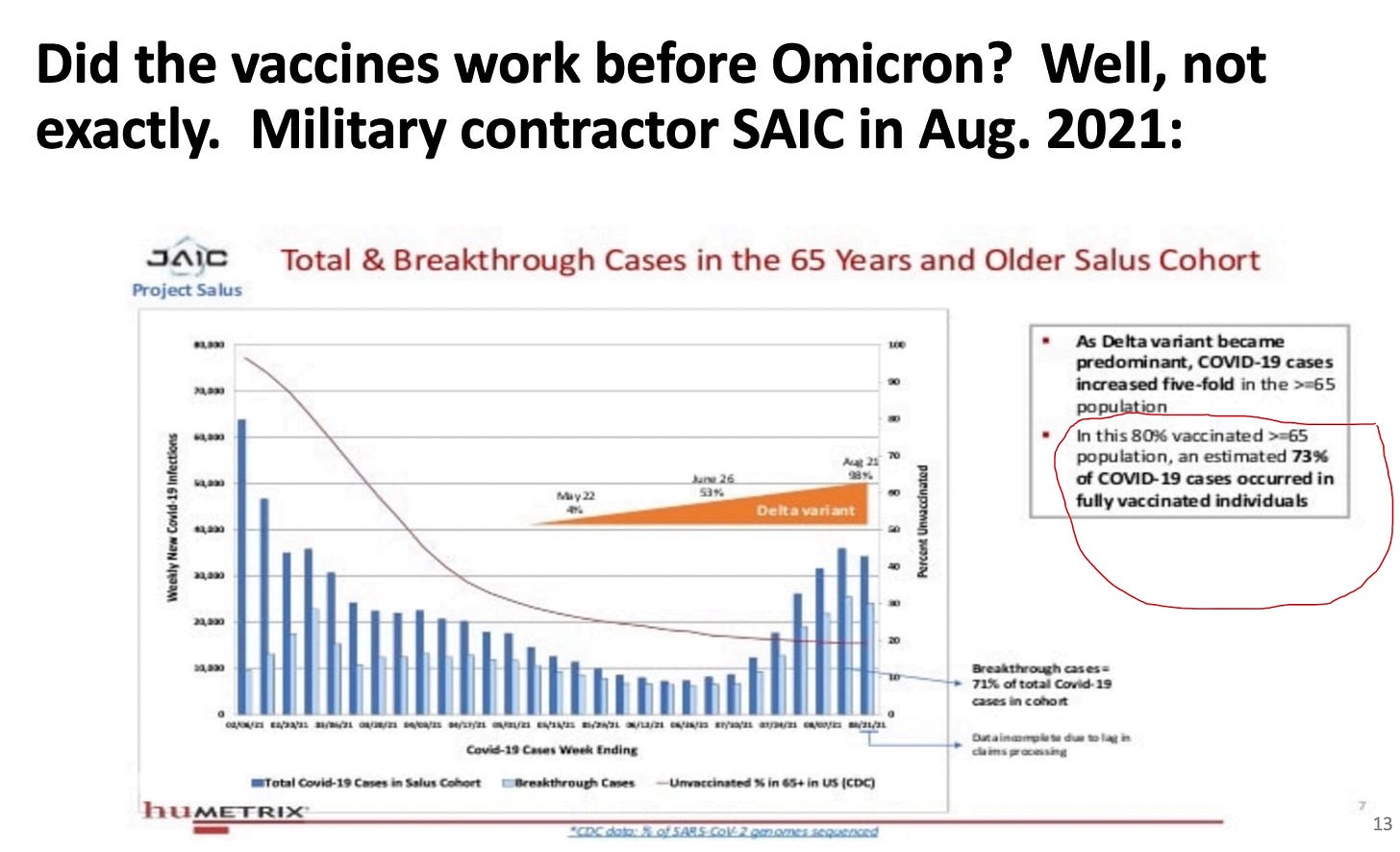
merylnass: Now Fleming-Dutra presents and gives the commercial that everyone should stay up to date with the vaccine and pregnant and breastfeeding women should especially get it. What? Her data claim the vaccine worked wonderfully for Delta. But here is some DOD contractor data on Delta in the vaccinated elders—about 10% benefit