A Bacterial Toxin Facilitating Chronic Infection
https://www.biozentrum.unibas.ch/news/detail/a-bacterial-toxin-facilitating-chronic-infection
A bacterial toxin facilitating chronic infection
Some pathogens persist in the body causing chronic infections. Researchers led by Prof. Christoph Dehio and Prof. Tilman Schirmer at the Biozentrum, University of Basel, have now discovered a mechanism of highly selective targeting of host proteins by a bacterial toxin that is critical for the bacteria to establish chronic infection. The study recently published in “PNAS” provides new insights into the activity and function of bacterial toxins.
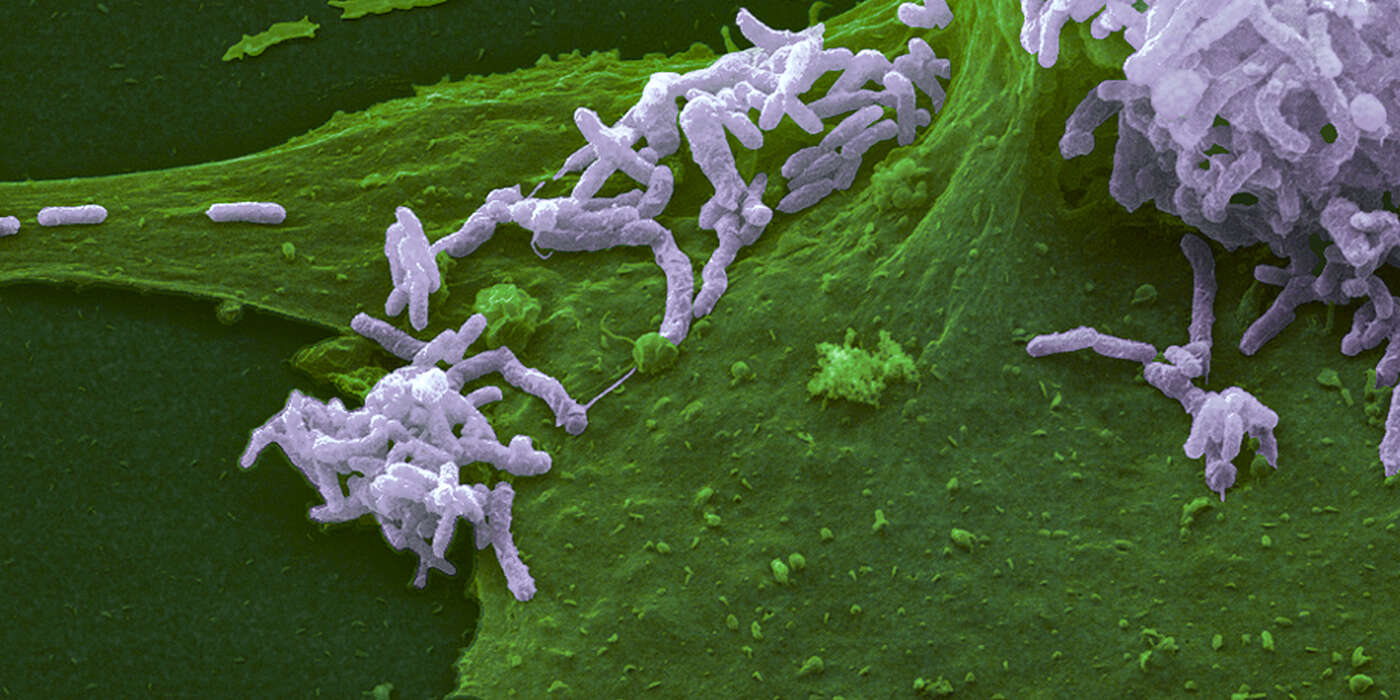
The bacterial pathogen Bartonella (purple) in interaction with human host cells (green).
When pathogens invade our body the immune system is put on alert. The body’s immune cells are recruited to the site of infection and an inflammatory reaction is initiated to rapidly eliminate the invaders. Some pathogens, however, have developed clever strategies to evade this line of defense. Bartonella is one of them. Manipulating the body’s cells to its advantage enables the pathogen to persist in the host.
Researchers led by Prof. Christoph Dehio and Prof. Tilman Schirmer at the Biozentrum, University of Basel, have now elucidated an important mechanism how Bartonella ensures long-term survival in the body. It injects a bacterial toxin into the body’s cells, which deactivates a specific group of proteins important for the immune response. This mechanism is vital for the pathogen’s capacity to cause chronic infection.
Bacterial toxins modulate signaling pathways in host cells
In mammalian cells, so-called Rho GTPases serve as molecular ON-OFF switches controlling a wide range of signaling pathways and thus pivotal cellular activities, such as cell movement, cytoskeletal dynamics and also the innate immune response. Due to their central regulatory role, this protein family is a target of choice for bacterial toxins. These thwart cellular signaling and facilitate the pathogen to survive in the host. However, many bacterial toxins targeting multiple GTPases cause massive collateral damage to the cells and typically limits pathogen survival to the acute infection phase.
Bartonella subtly colonizes the host
In contrast, Bartonella hijacks the host in a “gentle” way. The pathogen employs toxins that very selectively target host cell functions. In doing so, Bartonella reduces the efficacy of the immune system without causing collateral damage, enabling the pathogen to persist in the host. “We have now been able to elucidate the mechanism of highly selective recognition of specific host proteins by the Bartonella toxin Bep 1,” says Dehio. “Bep 1 exclusively targets proteins of the Rac-subfamily but not the other members of the large Rho GTPase family that are typically inactivated all together by toxins with primary acute infection patterns.”
Elucidating bacterial toxin selectivity
Employing a combination of structural analysis, modeling and biochemical methods, the researchers have now been able to elucidate the mechanism underlying this unique target selectivity. “The spectrum of target proteins is determined by large on shape complementarity and the electrostatic interactions of a short structural element in Bep1 with two protein segments unique to the Rac-subfamily,” explains Dehio. This simple, yet elegant, evolutionary treat equips Bartonella with a precise molecular tool to selectively interfere with host signaling.
Original publication:
Nikolaus Dietz, Markus Huber, Isabel Sorg, Arnaud Goepfert, Alexander Harms, Tilman Schirmer and Christoph Dehio. Structural basis for selective AMPylation of Rac-subfamily GTPases by Bartonella effector protein 1 (Bep1). Proceedings of the National Academy of Sciences (PNAS) 2021
Contact: Communications, Katrin Bühler
_____________
**Comment**
This right here, is why patients remain sick. We are typically filled with multiple stealth pathogens with the capability to quietly impede the normal mechanisms the body uses to clear infections. Sadly, Bartonella is not the only organism capable of this guerrilla warfare – Borrelia, the causative agent of Lyme disease also changes its outer surface protein to remain cloaked and accepted by the immune system. If antimicrobials are used, it also has the ability to shape shift and go into a dormant state only to reemerge when conditions are conducive for growth.
When will mainstream medicine get the memo?
For more:
- https://madisonarealymesupportgroup.com/2025/02/15/complex-role-of-bartonella-in-chronic-illness-1/
- https://madisonarealymesupportgroup.com/2025/03/07/treating-bartonella-medical-detective-pts-1-5/
- https://madisonarealymesupportgroup.com/2016/01/03/bartonella-treatment/
- https://madisonarealymesupportgroup.com/2025/05/21/bartonella-and-babesia-detected-in-patients-with-chronic-illness/
- https://madisonarealymesupportgroup.com/2025/10/22/explaining-dddct-protocol-for-treating-bartonella-parts-1-5/
- https://madisonarealymesupportgroup.com/2024/05/09/the-mind-blowing-truth-about-bartonella-dr-ed-breitschwerdt/
- https://madisonarealymesupportgroup.com/2022/01/18/clinical-spectrum-of-persistent-bartonella-infection-and-considerations-in-diagnosis-treatment/
- https://madisonarealymesupportgroup.com/2019/02/27/advanced-imaging-found-bartonella-around-pic-line/
- https://madisonarealymesupportgroup.com/2021/05/20/chronic-bartonella-infections-in-animals-and-humans-crypto-infections-conference/


