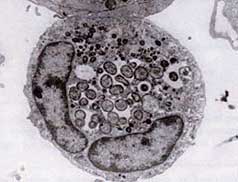
Ultrastructure of A.phagocytophilum by transmission
electron microscopy. Photo by V.Popov, reprinted
from Dumler JS et al. Human granulocytic
anaplasmosis and Anaplasma phagocytophilum.
Emerg Infect Dis;11:1828-34.
http://www.health.state.mn.us/divs/idepc/diseases/anaplasmosis/hcp.html
Human anaplasmosis (HA), formerly known as human granulocytic ehrlichiosis (HGE), is a small, obligate, gram-negative bacterial disease that is unusual in its tropism to neutrophils, and is caused by Anaplasma phagocytophilum, a rickettsial bacterium. It was first recognized in 1990, when a western Wisconsin patient developed a severe febrile illness following a tick bite and died two weeks later; however, it has been known to cause disease in animals since 1932. Human ehrlichiosis, a similar disease, is caused by Ehrlichia chaffeensis and is found throughout much of southeastern and south-central United States. Another related form of ehrlichiosis caused by the Ehrlichia muris-like agent was identified in Minnesota and Wisconsin patients in 2009. The median age of patients with HGA is around 50 years old. Over 4000 total cases have been reported in the CDC’s Morbidity and Mortality Weekly since the disease became nationally reportable; as with most tick-borne diseases, the true incidence is suspected to be considerably higher.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2882064/ Cases occur year-round, with a peak incidence during June and July, perhaps reflecting the shorter arthropod season in these northern states or the relative importance of the nymphal stage of Ixodes ticks in disease transmission. Given the ubiquity of the tick vector, it is not surprising that cases of HGA have been confirmed world-wide, including Europe and Asia (China, Siberian Russia, and Korea).
In dogs, persistent infection has been reported to last over 10 years and in the absence of treatment can persist for the life of the dog. Other strains persist for months and then are naturally cleared.
Transmission:
http://www.health.state.mn.us/divs/idepc/diseases/anaplasmosis/hcp.html
http://www.capcvet.org/capc-recommendations/ehrlichia-spp-and-anaplasma-spp1/ Anaplasmosis is known to be transmitted to humans by Ixodes scapularis (blacklegged tick or deer tick), the same tick that transmits Lyme disease (borrelia). It can also be transmitted via blood transfusion and contaminated needles or surgical instruments.
Symptoms:
Onset of illness occurs 5 to 21 days after exposure to an infected tick. Infection can range from asymptomatic infection to fatal disease. Common signs and symptoms include fever (often over 102°F), chills, headache, and myalgias. Nausea, vomiting, anorexia, acute weight loss, abdominal pain, cough, diarrhea, and change in mental status are reported less frequently. Highly suggestive laboratory findings include leukopenia – a decrease in white blood cells making you more susceptible to infection (WBC< 4,500/mm³), thrombocytopenia – a decrease in platelets causing bruising and bleeding (platelets <150,000/mm³), and increased aminotransferase levels – relating to liver damage. Most of the damage it causes appears to be related to host inflammatory processes, as there is little evidence of a correlation between the number of organisms and host disease severity.
http://www.columbia-lyme.org/patients/tbd_ehrli-anapla.html
Compared with HME (human monocytic ehrlichiosis), HGA (human granulocytic anaplasmosis) appears less likely to involve the central nervous system, but peripheral neuropathies are more common and can last weeks to months. Among the neurologic findings reported in the medical literature are facial palsy, demyelinating polyneuropathy and brachial plexopathy. Respiratory distress syndrome and a septic or toxic shock-like syndrome have been reported, but appear to be less common than in HME. The overall fatality rate from HGA also seems to be slightly lower than that of HME, with most of the deaths resulting from opportunistic infections (for example, herpes simplex esophagitis, Candida pneumonitis, and pulmonary aspergillosis) in immunocompromised patients.
Unusual presentations may be the result of coinfections with Borrelia burgdorferi (Lyme disease agent) and/or Babesia microti (babesiosis agent), as a single feeding tick may transmit multiple disease agents.
Cases of HA acquired through blood transfusions have been documented. Include HA in the rule-out for patients who develop a febrile illness with thrombocytopenia following blood transfusion. Suspected transfusion-associated anaplasmosis should be reported.
Prevalence:
A large survey by Lymedisease.org found that 53% stated they had coinfections and 30% responded they had two or more coinfections. Similar results were found in Canada. While the most common coinfections found in Canada were Bartonella (36%) and Babesia (19%), Anaplasma took third place (13%). https://www.lymedisease.org/lymepolicywonk-study-finds-coinfections-in-lyme-disease-common-2/ Here in the U.S. the results showed that 5% of patients with Lyme also had Anaplasmosis; however, please realize most doctors are not looking for this, the testing is poor, many states don’t require reporting and many cases go unreported. In 2014, there were 2800 confirmed cases of HA. Rhode Island, Minnesota, Connecticut, Wisconsin, New York and Maryland are the hotbeds. https://www.cdc.gov/mmwr/volumes/65/rr/pdfs/rr6502.pdf
Master Herbalist, Stephen Buhner, in his book Natural Treatments for Lyme Coinfections (Anaplasma, Babesia, and Ehrlichia states on page 24, “In other words, if you want to successfully treat someone who is infected with a vector-borne infection you need to realize up front that it is usually the case that coinfection has occurred and you have to look at the interactive picture, not merely single infectious agents.” Demonstrating another unique interplay, one study found that ticks express an antifreeze type substance to enhance survival when they are infected with Anaplasma. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2929727/ In the world of microbes there is definitely a “You scratch my back, I’ll scratch yours.”
Tests:
http://www.columbia-lyme.org/patients/tbd_ehrli-anapla.html
Standard blood tests in HGA usually reveal findings similar to those seen in HME: leukopenia, thrombocytopenia and liver function abnormalities (elevated transaminases). However, the hematological abnormalities frequently resolve by the second week of symptoms, so their absence should be interpreted in that context if patients are presenting later in the course of their illness. In general, empiric antibiotic treatment should be considered for patients in endemic areas who present with an acute febrile illness suggestive of HGA. For specific diagnosis, Wright or Giemsa-stained blood smears have a slightly higher yield than with HME, but are still not optimal for general clinical utility, given that there appears to be a wide variation (25-75%) in the sensitivity of these tests in visualizing morulae in host neutrophils. More helpful, but not always available, are polymerase chain reaction (PCR) tests, which are estimated to have a sensitivity of 67-90%. Prior antibiotic therapy dramatically reduces the sensitivity of both of these diagnostic methods. Serologic testing is useful to confirm the diagnosis of anaplasmosis. The most commonly used method is indirect immunofluorescence (IFA) of IgM and IgG anti-A. phagocytophilum antibodies. Seroconversion is perhaps the most sensitive laboratory evidence of A. phagocytophilum infection, but is not always obtained in a timely enough manner to provide useful input on clinical (i.e., treatment) decisions.
According to the CDC:
Any two of the following three tests for evidence of infection with Anaplasma phagocytophilum are recommended:
*An indirect immunofluorescence assay (IFA) is the principal test used to detect HA infection. Acute and convalescent phase serum samples can be evaluated to look for a four-fold change in antibody titer to A. phagocytophilum.
*Intracellular inclusions (morulae) also may be visualized in granulocytes on Wright- or Giemsa- stained blood smears.
*Polymerase chain reaction (PCR) assays are being used increasingly to detect A. phagocytophilum DNA.
According to animal studies, morulae are usually difficult to find in blood smears, even during the acute stage of disease. It is also stated that serology may be helpful in identifying antibodies but may not detect early infections during the acute phase of disease.
 Courtesy of Lymestats.org
Courtesy of Lymestats.org
Treatment: Discuss all treatments with your health care professional
http://www.columbia-lyme.org/patients/tbd_ehrli-anapla.html
The optimal dose and duration of antibiotic treatment for anaplasmosis has not been definitively established, but it is clear that A. phagocytophilum is highly sensitive to tetracyclines. Thus, oral doxycycline is the recommended treatment, at the same dose used for Ehrlichia infections: 200 mg/day in two divided doses. The usual treatment duration is 5-10 days, which is extended if there is suspected coinfection with B. burgdorferi, the agent of Lyme disease. In any case, treatment should continue for at least three days after the patient’s fever resolves. Response to treatment is usually rapid; if the patient remains febrile more than two or three days after initiation of doxycycline therapy, the diagnosis should be revisited.
As with Ehrlichia infections, rifampin is used in cases where doxycycline is contraindicated, such as pregnancy or allergy. Rifampin has also been used successfully in pediatric cases, and thus is sometimes employed in mild cases of pediatric A. phagocytophilum infection. If coinfection with B. burgdorferi is suspected in a pediatric case, doxycycline is sometimes used as an initial treatment for 3-5 days, with another antibiotic employed thereafter to complete the somewhat longer recommended treatment period for early Lyme disease.
Treatment according to the CDC: (Notice the dose is lowered)
http://www.cdc.gov/rmsf/doxycycline/index.html
HA patients typically respond dramatically to doxycycline therapy (100 mg twice daily until the patient is afebrile for at least 3 days). Other tetracycline drugs also are likely to be effective. In general patients with suspect HA and unexplained fever after a tick exposure should receive empiric doxycycline therapy while diagnostic tests are pending, particularly if they experience leukopenia and/or thrombocytopenia.
http://www.jpeds.com/article/S0022-3476(15)00135-3/pdf?ext=.pdf
According to the CDC, Doxycycline is the most effective antibiotic for the treatment of suspected rickettsial infections for all ages, including Rocky Mountain Spotted Fever, and delay in treatment may lead to severe illness and/or death. Children are five times more likely than adults to die from RMSP and a new study found that short courses of doxy can be used in children without causing tooth staining or weakening of tooth enamel. Prior to this, doctors were reticent using Doxy due to studies in the 50’s showing a link between Tetracycline (binds to calcium) use in young children and tooth weakening and staining. Doxy, a newer medication, binds to calcium less readily and, according to the CDC, if used in the correct dose and duration for rickettsial diseases, should cause no harm. It’s also the treatment of choice according to the American Academy of Pediatrics (AAP). Doxycycline treatment should be continued for at least 3 days after fever resolves, and the usual duration of therapy is 7-10 days. Chloramphenicol is considered a second-line therapeutic agent, as it is significantly less effective at preventing fatal outcome; other broad-spectrum antimicrobial agents typically used to treat sepsis are not effective at preventing fatal outcome due to RMS.
http://www.ilads.org/lyme/what-to-do-if-bit-by-tick.php
According to ILADS (International Lyme and Associated Diseases Society) Doxycycline has the advantage of treating numerous tick borne illnesses such as Lyme (borrelia), Ehrlichia, Anaplasma, Q Fever, and Rocky Mountain Spotted Fever. They state the downside is that Doxy causes significant sun sensitization, can be hard on the stomach, and the usual dosing may not reach therapeutic levels.
Recent data suggests that treatment may not clear organisms in animals.
If you find a doctor willing to become properly educated on tick borne illness, please give them this link: https://madisonarealymesupportgroup.com/2017/06/20/help-doctors-get-educated-on-lyme-and-tick-borne-illness/



 Courtesy of
Courtesy of