Babesia Treatment: Dr. Cameron’s Clinical Tips
https://danielcameronmd.com/babesiosis-treatment/
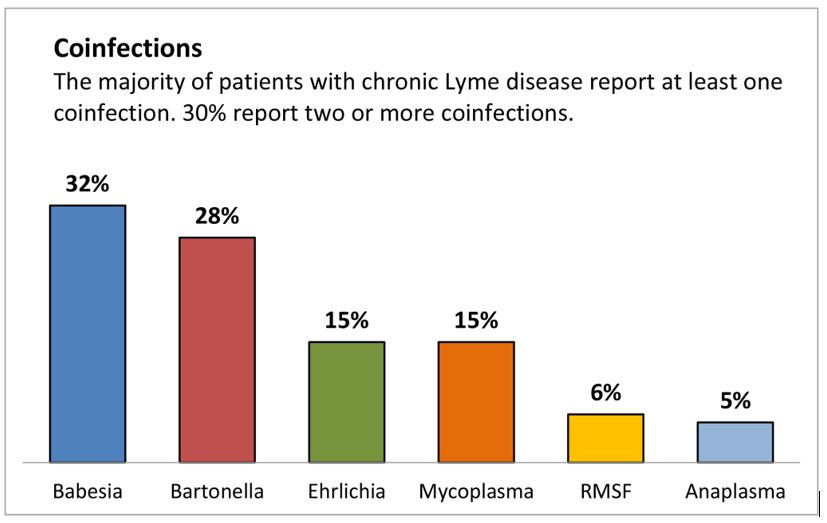
Babesiosis treatment is often overlooked—especially in patients who also have Lyme disease. Babesiosis is a malaria-like illness caused by microscopic parasites that infect red blood cells. It’s frequently found alongside Lyme, but just as often, it’s missed.
Unlike Lyme, which is caused by the bacterium Borrelia burgdorferi, babesiosis is caused by parasites (Babesia microti or Babesia duncani) and does not respond to doxycycline. Yet I continue to see Lyme patients who are only treated with doxycycline—even when symptoms strongly suggest babesiosis may be present.
So what do I use when babesiosis testing is negative but suspicion is high?
Let’s walk through my approach.
Why Testing for Babesiosis Falls Short
Babesiosis tests often fail—especially in patients with chronic or relapsing symptoms. Here’s why:
-
-
Parasites are visible on blood smear only in early infection
-
PCR (detects DNA) may miss low-level infections
-
Antibody tests may remain negative for months—or fade over time
-
In my practice, I increasingly see positive Babesia antibodies after months of symptoms, even when earlier testing was negative. This tells me two things:
-
- Symptoms often precede test positivity.
- Waiting for test confirmation may delay needed treatment.
Babesiosis Treatment: Key Symptoms That Warrant Empiric Therapy
When testing fails but the clinical picture fits, I move forward with treatment.
Common babesiosis symptoms in my patients include:
-
-
Night sweats (often drenching)
-
Shortness of breath or “air hunger”
-
Fatigue that worsens with activity
-
Lightheadedness, POTS-like symptoms
-
Temperature dysregulation
-
Anxiety or depression out of proportion
-
These symptoms are sometimes dismissed as menopause, anxiety, or long COVID—but in a patient with a history of Lyme disease or tick exposure, they raise concern for babesiosis.
First-Line Babesiosis Treatment
When babesiosis is suspected, the first-line treatment I use is: Atovaquone + Azithromycin
This combination remains the most commonly prescribed regimen, with proven effectiveness.
-
-
Atovaquone is an anti-parasitic that targets the Babesia organism in red blood cells.
-
Azithromycin is an antibiotic that complements atovaquone’s activity.
-
Practical Note:
Many clinicians prescribe Mepron® (atovaquone 750 mg/5 mL oral suspension), but I’ve had success using Malarone® (atovaquone 250 mg + proguanil 100 mg tablets), which:
-
-
Is better tolerated by many patients
-
Is easier to obtain in outpatient practice
-
Comes in pediatric-sized tablets (62.5 mg/25 mg) useful for dose titration
-
This flexibility allows me to individualize babesiosis treatment—especially for sensitive patients who cannot tolerate full adult dosing at first.
Newer Option: Tafenoquine
For resistant or relapsing babesiosis, I’ve also begun using: Tafenoquine (Krintafel®)
-
-
Originally approved for malaria
-
May be effective in difficult Babesia cases
-
Requires screening for G6PD deficiency before use
-
Still considered off-label in many outpatient Lyme protocols
-
Tafenoquine is not a first-line therapy, but it may have a role when patients relapse despite standard babesiosis treatment. I’m following the research closely.
Why I Don’t Use Clindamycin or Quinine
While clindamycin and quinine are sometimes recommended for severe babesiosis (especially hospitalized cases), I have not been using them in outpatient care. In my experience:
-
-
Clindamycin + quinine causes significant nausea, tinnitus, and other side effects
-
Not well tolerated in chronic or relapsing Lyme patients
-
Alternative regimens (like atovaquone-based therapies) are typically sufficient
-
If a patient does not respond to first-line babesiosis treatment, I evaluate for possible co-infections (like Bartonella or Ehrlichia), medication tolerance, and drug absorption before moving to more aggressive regimens.
What I Watch for During Treatment
When treating babesiosis, I monitor:
-
-
Liver enzymes (especially with Mepron)
-
Hemoglobin and hematocrit (to assess for hemolysis)
-
Symptom patterns (including Herxheimer reactions)
-
Drug tolerance and adherence
-
Improvement can take time. But when patients begin to regain energy, lose their night sweats, and tolerate light exertion again, it’s a sign that treatment for babesiosis is working.
Final Thoughts on Babesiosis Treatment
Babesiosis doesn’t always show up on a lab test—but it can still cause profound illness, especially in those with Lyme disease or immune dysfunction.
If you’ve been treated for Lyme disease but still suffer from:
-
-
Fatigue
-
Night sweats
-
Breathlessness
-
POTS-like symptoms
-
… don’t assume it’s “just Lyme.” Babesiosis treatment may be the missing piece.
References
-
-
-
- Babesia symptoms often missed in chronic Lyme disease patients
https://danielcameronmd.com/treating-babesia-negative-tests/
-
Vannier E, Krause PJ. Human babesiosis. New England Journal of Medicine. 2012;366(25):2397–2407.
https://www.nejm.org/doi/full/10.1056/NEJMra1202018 -
Wormser GP, Dattwyler RJ, Shapiro ED, et al. Clinical assessment, treatment, and prevention of Lyme disease, anaplasmosis, and babesiosis. Clinical Infectious Diseases. 2006;43(9):1089–1134.
https://academic.oup.com/cid/article/43/9/1089/422463 -
Krause PJ, Auwaerter PG, Bannwarth M, et al. Babesiosis. Infectious Disease Clinics of North America. 2015;29(2):357–370.
https://pubmed.ncbi.nlm.nih.gov/25999229/
- Babesia symptoms often missed in chronic Lyme disease patients
-
-
________________
**Comment**
Fantastically practical article.
For more:
- https://madisonarealymesupportgroup.com/2016/01/16/babesia-treatment/
- https://madisonarealymesupportgroup.com/2022/03/24/kill-babesia-treatment-guide-update/
- https://madisonarealymesupportgroup.com/2024/01/08/tafenoquine-atovaquone-achieves-radical-cure-confers-sterile-immunity-in-experimental-models-of-human-babesiosis/
- https://madisonarealymesupportgroup.com/2022/04/08/tafenoquine-for-relapsing-babesiosis/
- https://madisonarealymesupportgroup.com/2022/07/01/relapsing-babesia-treated-successfully-with-tafenoquine/
- https://madisonarealymesupportgroup.com/2025/05/21/bartonella-and-babesia-detected-in-patients-with-chronic-illness/
- https://madisonarealymesupportgroup.com/2025/05/07/one-unit-of-blood-led-to-babesia-infection/
- https://madisonarealymesupportgroup.com/2021/01/08/rising-geriatric-babesia-cases-may-require-longer-treatment/
- https://madisonarealymesupportgroup.com/2020/05/12/babesia-infection-3-weeks-after-lyme-treatment/ I must add that this our experience as well: we didn’t notice the Babesia until weeks if not months after we started treatment for Lyme disease.
- https://madisonarealymesupportgroup.com/2024/02/05/babesia-with-dr-cameron/
I believe Dr. Horowitz states that when you start treatment for Babesia, you need to keep going until it’s gone. Stopping and starting makes it harder to treat in the long run.