NEW YORK, NY (March 30, 2021)–A new center that will provide specialized care for patients with Lyme disease and other tick-borne diseases will begin seeing patients this summer at Columbia University’s Vagelos College of Physicians and Surgeons.
The Cohen Center for Health and Recovery from Tick-Borne Diseases, supported by a $16 million gift from the Steven & Alexandra Cohen Foundation, will be the first in New York City to offer dedicated treatment for people with Lyme and related diseases.
The foundation’s gift will also fund a national clinical trials network that will focus on identifying more effective treatments for patients with Lyme and tick-borne diseases.
“There is a growing need for evidence-based treatments for patients with Lyme and other tick-borne diseases,” said Anil Rustgi, MD, interim executive vice president and Dean of the Faculties of Health Sciences and Medicine at Vagelos College of Physicians and Surgeons and Columbia University Irving Medical Center. “This generous gift from the Cohen Foundation will allow us to build on our faculty’s expertise in Lyme and tick-borne disease patient care and research, providing access to comprehensive, coordinated treatment.”
“We know firsthand the devastation that Lyme and tickborne diseases can cause, and we are thrilled to support this innovative center and clinical trials network to help bring treatments and hope to patients and their families,” said Alex Cohen, President, Steven & Alexandra Cohen Foundation.
Growing Need for Lyme Disease Experts and Research
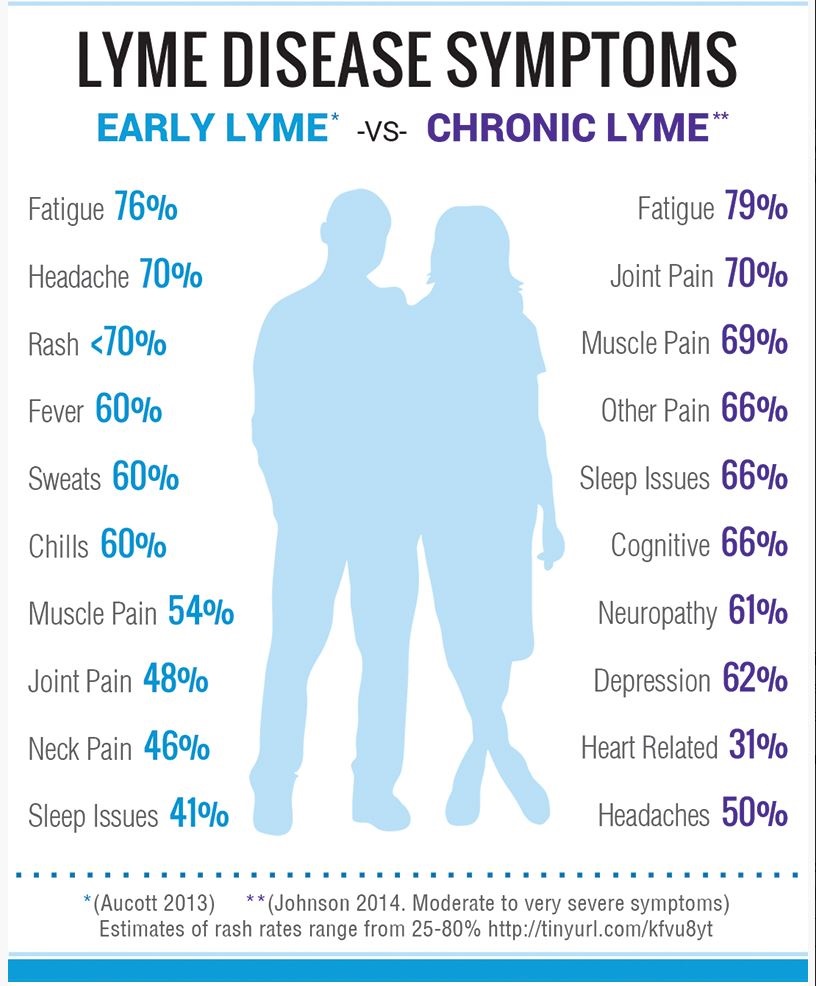
The estimated incidence of Lyme disease in the United States has been steadily rising, to approximately 476,000 new cases (including individuals who are newly diagnosed or treated for the disease) annually. Despite antibiotic treatment, approximately 10-20% of these new infections will lead to distressing and potentially disabling symptoms, such as severe fatigue, joint and muscle pain, neurologic symptoms, and cognitive problems that may last for months or years.
There is limited expertise in treating the acute and chronic aspects of tick-borne diseases, making it difficult for patients to find high-quality, specialized care. In addition, little research has been done to determine which treatments are most effective for persistent symptoms, which can interfere with daily activities and work.
“Like COVID-19 ‘long-haulers,’ many people with tick-borne diseases were completely well until their infection precipitated a cascade of chronic, multi-system effects,” says Brian Fallon, MD, director of the Cohen Center for Health and Recovery from Tick-Borne Diseases. “A comprehensive evaluation takes time. In addition, the cost of care for patients with chronic symptoms can be prohibitive, requiring multiple visits to physicians who may not be aware of the latest research on tick-borne illnesses. Our center will be the first to address all of these issues by offering access to affordable care with experienced physicians while at the same time integrating research and physician training into our clinical model.“
Fallon plans to hire additional clinicians to begin seeing patients via telemedicine in July. New clinicians will include family, integrative, and behavioral medicine specialists. Patients with acute or chronic symptoms are expected to be able to see the Cohen Center team this fall in the center’s new clinical space in the Neurological Institute.
“Many patients with chronic symptoms related to tickborne illness are misdiagnosed with conditions such as depression, chronic fatigue syndrome, or even psychosomatic disorder,” says Shannon Delaney, MD, co-director of the Cohen Center. “Children infected with a tick-borne illness may have dramatic and disabling neurologic or psychiatric symptoms that seem to occur overnight.”
Cohen Center patients will be invited to enroll in clinical trials, as well as brain imaging studies and neuropsychiatric studies. Studies that identify biologic markers can help determine treatment approaches.
The Cohen Center will provide training for family medicine fellows and medical students on how to evaluate and treat patients with tick-borne diseases.
“Our educational component will allow us to create a pipeline of family medicine and primary care physicians with experience in caring for patients with the unique and varied set of symptoms associated with tick-borne diseases,” says Krishna Desai, MD, assistant professor of medicine at Columbia University Vagelos College of Physicians and Surgeons and director of family and integrative medicine at the center.
First Clinical Trials Network for Lyme and Tick-borne Diseases
The Cohen gift expands Lyme and tick-borne diseases research by establishing the first clinical trials network for multi-site clinical trials and pilot studies to assess treatments. Columbia will be the coordinating site for the trials. Experts at Johns Hopkins University School of Medicine and Children’s National Hospital are key members of the national network.
Academics, community physicians, and the general public will be invited annually to submit treatment study ideas to the clinical trials network for consideration.
“There’s a critical need for effective therapies for patients with Lyme and tick-borne diseases,” says Fallon. “The clinical trials network will be a powerful engine to drive high-quality research in tick-borne diseases, including large-scale clinical trials and potentially transformational early stage research.”
###
More information
Brian Fallon, MD, MPH, is professor of clinical psychiatry at Columbia University Vagelos College of Physicians and Surgeons and a physician-scientist with expertise in disorders intersecting medicine, neurology, and psychiatry. He is co-author of “Conquering Lyme Disease” with Dr. Jenifer Sotsky (Columbia University Press, 2017). He is also director of the Lyme and Tick-Borne Diseases Research Center at Columbia University.
Shannon Delaney, MD, is assistant professor of psychiatry at Columbia University Vagelos College of Physicians and Surgeons and an expert in child and adult neuropsychiatry and infection-triggered neuropsychiatric disorders.
Krishna Desai, MD, is assistant professor of medicine at Columbia University Vagelos College of Physicians and Surgeons.
The clinical trials network collaborating investigators include John Aucott, MD, Director of the Johns Hopkins Lyme Disease Research Center, in Baltimore, Maryland, and Roberta DeBiasi, MD, Chief, Division of Pediatric Infectious Diseases, Children’s National Hospital in Washington, D.C.
Columbia University Irving Medical Center provides international leadership in basic, preclinical, and clinical research; medical and health sciences education; and patient care. The medical center trains future leaders and includes the dedicated work of many physicians, scientists, public health professionals, dentists, and nurses at the Vagelos College of Physicians and Surgeons, the Mailman School of Public Health, the College of Dental Medicine, the School of Nursing, the biomedical departments of the Graduate School of Arts and Sciences, and allied research centers and institutions. Columbia University Irving Medical Center is home to the largest medical research enterprise in New York City and State and one of the largest faculty medical practices in the Northeast. For more information, visit cuimc.columbia.edu or columbiadoctors.org.
About the Steven and Alexandra Cohen Foundation
The Steven & Alexandra Cohen Foundation is committed to inspiring philanthropy and community service by creating awareness, offering guidance, and leading by example to show the world what giving can do. The Foundation’s grants support nonprofit organizations based in the United States that either help people in need or solve complex problems. The Foundation also spearheads grassroots campaigns to encourage others to give. For more information, visit http://www.steveandalex.org.