https://www.galaxydx.com/the-basics-of-serology/
written on
The Basics of Serology
Reading a diagnostic company’s menu can often feel like interpreting another language. The commonly used name of an infection may not be listed, and the scientific name of the pathogen may not be familiar (e.g., Lyme disease vs. Borrelia burgdorferi).
To make things more difficult, acronyms for the testing methods can seem like randomly grouped letters for those of us who are not researchers or do not work in a clinical setting. Serology, an umbrella term for testing serum, is a common method that Galaxy Diagnostics customers frequently contact us for help deciphering.
The most basic definition of serology is “the study of blood serum”. Blood serum is created when clotted blood is collected in a serum-separator tube and then spun in a centrifuge at high speed. This forces red and white blood cells through a gel matrix at the bottom of the tube, leaving a clear to yellowish liquid called serum. The components that remain include electrolytes (sodium, potassium, etc.), hormones, and an assortment of proteins.
Immunoglobulins (Ig), or antibodies, can be found in serum and are useful for vector-borne disease testing. They are proteins created by the immune system when a foreign substance invades the bloodstream. The development of these proteins may be delayed, suppressed or altered if a person’s immune system is different for a variety of reasons, including the use of immune suppressing medications or the presence of an autoimmune condition such as lupus.
The foreign substance, called an antigen, could be a wide variety of things, such as a toxic by-product of pathogen metabolism, a protein located on the surface of the bacteria itself, or a virus. White blood cells, specifically B cells, bind antigens and create unique antibodies that help mount a strong immune response.
In humans, typical antibodies are shaped like a “Y” and have two binding sites that are specific to an antigen. As they float around the bloodstream, they can bind the antigens that they were created to match. Once an antigen is bound, it may be broken down immediately or another immune cell, such as a T-cell, may be signaled to destroy it. In a sense, antibodies act as the whistle blowers of the immune system.
There are five classes of antibodies:
- IgA
- IgE
- IgD
- IgM
- IgG
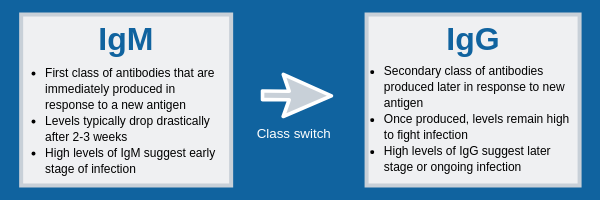
Antibodies that are relevant for vector-borne serology testing are IgM and IgG. However, IgM only make up about 5-10% of the total blood serum, whereas IgG make up about 75-80%. So why test for both?
Generally, when a new antigen is encountered, B-cells produce about six times more IgM than IgG. IgM are the largest antibodies in humans and have more antigen binding sites than IgG. Therefore, they mount a strong initial defense and can provide laboratory evidence of a new infection.
However, they have some difficulties in a test tube. Some research shows that IgM can be less reliable than IgG due to increased cross-reactivity with non-target antigens. For example, researchers in one study found that 45% of samples tested positive for both Borrelia burgdorferi and Yersinia species on an IgM Western blot assay created for Yersinia specifically. Yersinia species include the pathogen that causes plague. Imagine being told that you had plague when you really had Lyme disease, yikes!
IgG can be a little more useful in the lab. They provide a secondary response that helps rid the body of the remaining infection. Generally, about 30 days after the initial infection, the concentration of IgM decreases drastically and IgG concentration increases.
IgG antibodies are typically more specific and provide a long-term defense, remaining in the bloodstream for weeks to months. Therefore, if the host is exposed to the same antigen in the future, the immune system can recognize and eliminate it quicker. Since IgG antibodies can remain in the body for so long, it is difficult to interpret the results of IgG tests without the context of patient medical history and current symptoms.
Molecular evidence of infectious pathogens (such as PCR testing to identify pathogen DNA) is often very difficult to detect, so many diagnostic methods rely on the antibodies created by the host’s immune system to rule out infections. A few common examples of these “immunoassays” are the ELISA, IFA, and Western blot.
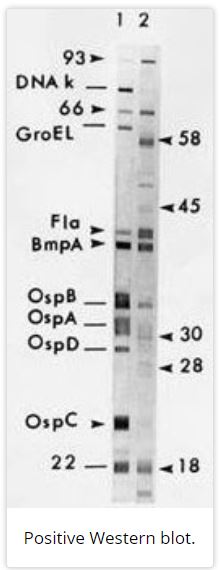
The ELISA (Enzyme-Linked Immunosorbent Assay) and IFA (immunofluorescent assay) rely on chemical reactions that occur when antibodies present in a sample interact with antigens prepared by the lab. If there are antibodies present, a signal will be produced that can be seen by an automated device or by lab personnel. For example, in “reactive” IFA samples, a green fluorescence is measured in samples at different dilutions to determine the concentration of antibodies present. Likewise, Western blot (or sometimes referred to as immunoblots) look for the IgM and IgG themselves that are present after infection occurs. The serum sample is prepared and run through a gel matrix using an electrical current, transferred via a “blotting” technique, and then stained so “bands” become visible. This process separates proteins via size so that the person reading the blot can determine the presence of antibodies based on known markers. According to CDC criteria, a person is positive for Lyme disease on Western blot if there are 2 out 3 IgM or 5 out of 10 IgG diagnostic bands present.
Serology testing is a secondary method of testing for a pathogen. The body’s immune system response can amplify signals about the presence of pathogens that are easier to capture in a laboratory. However, those signals themselves can be difficult to interpret as they are not always the same and can indicate different things about when – and if – a pathogen has been encountered by the body’s immune system.
References
Wine, Y. et al. (2015). Serology in the 21st century: The molecular-level analysis of the serum antibody repertoire. Current Opinion in Immunology, 35, 89-97. doi:10.1016/j.coi.2015.06.009 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4553097/pdf/nihms709220.pdf
Maggi, R. G. et al. (2014). Comparison of serological and molecular panels for diagnosis of vector-borne diseases in dogs. Parasites & Vectors, 26(7), 127. doi:10.1186/1756-3305-7-127 https://www.ncbi.nlm.nih.gov/pubmed/24670154
Rawlins, M. L. et al. (2005). Evaluation of a Western blot method for the detection of Yersinia antibodies: Evidence of serological cross-reactivity between Yersinia outer membrane proteins and Borrelia burgdorferi. Clinical and Diagnostic Laboratory Immunology, 12(11), 1269-1674. doi:10.1128/CDLI.12.11.1269-1274.2005 https://www.ncbi.nlm.nih.gov/pubmed/16275939
Centers for Disease Control and Prevention. (2019). Lyme disease. Available at: https://www.cdc.gov/lyme/diagnosistesting/index.html
__________________
For more: https://madisonarealymesupportgroup.com/2018/09/12/lyme-testing-problems-solutions/
https://madisonarealymesupportgroup.com/2017/08/15/reliability-of-lyme-testing/
https://madisonarealymesupportgroup.com/2018/09/08/whats-the-best-test-for-lyme-dr-rawls/
About testing he states that the Western Blot appears to be the most positive finding in clinical LD, but…“setting arbitrary level of antibodies to diagnose a disease that has not been amenable to Koch’s postulates seems open to question. By the same token, ignoring antibody results unless they meet arbitrary levels seems suspect. The vast majority of patients in this series showed some WB antibody exposure, but many did not meet the arbitrary limits set….in our present state of knowledge, the diagnosis of chronic Lyme disease is a clinical one. Many of the patients in this series have suffered serious ‘hurts’ when they have been told that they could not have LD because their WB did not meet arbitrary limits.”- Dr. Waisbren