https://www.wiscontext.org/confounding-geography-lyme-disease-us
The Confounding Geography Of Lyme Disease In The U.S.
Scientists Explore How Regional Variations In Tick Behavior Drives Difference In Infections
Scott Gordon
WisContext
June 6, 2018

Graham Hickling/University of Tennessee
The Upper Midwest and the northeastern regions of the United States are increasingly a carpet of Lyme disease cases each summer and autumn. But the southeastern part of the country — a vast expanse of hot and humid territory and certainly hospitable to the ticks that carry Lyme-causing bacteria — gets off relatively easy. At times, the question of whether Lyme disease even exists in the South has spurred fierce debate among entomologists and public-health officials. In short, yes, it does exist in the South, but is far less prevalent than in points north.
The medical community officially began to recognize Lyme disease in the 1970s. It’s named after Lyme, Connecticut, where residents became convinced they were dealing with a little-understood disease and not getting the proper diagnosis or treatment. In the early 1980s, a medical entomologist named Willy Burgdorfer discovered that Lyme was a bacterial infection — which is why the main Lyme-causing bacteria in North America is called Borrelia burgdorferi — and that deer ticks, also known as black-legged ticks (Ixodes scapularis), were spreading it. Deer ticks were first discovered in Wisconsin in 1965 but became more common around the state during the 1980s. Since that time, Lyme disease diagnoses have increased dramatically in the Northeast and Upper Midwest, but much less so in the southern or the western U.S.
While the nation as a whole is seeing an increase in insect-borne illnesses, the northern U.S. still gets the brunt of Lyme season. Entomologists and epidemiologists are still trying to piece together what exactly drives this stark regional difference.
Questing for answers
North America’s deer ticks exhibit different behaviors in different regions, and have evolved into genetically distinct populations of the same species. Northern deer ticks have exhibited more outgoing behavior than their southern counterparts, said Jean Tsao, a disease ecology professor at Michigan State University.
Tsao, who studies ticks across the Midwest and the country, co-led a study that observed tick nymphs from Wisconsin, Rhode Island, Tennessee and Florida in an artificial habitat. The scientists looked closely at what’s known as “questing” behavior — that is, how ticks go about looking for a host animal to feed on. The ticks from Rhode Island and Wisconsin spent a lot more time climbing up the walls and the dowels meant to simulate the stems of plants. However, the Tennessee and Florida ticks tended to stay under the “leaf litter,” a layer of dead vegetation that settles on the ground in forests or other plant-dense habitats.

Graham Hickling/University of Tennessee/PLos One
In an experiment conducted in an artificial habitat, black-legged ticks from Wisconsin and Rhode Island were more likely to ascend dowels to “quest” for prey.
Both the northern and southern ticks are looking for hosts — they all have to imbibe blood and become grotesquely engorged to progress from one stage of life to the next. The ticks can pick up bacteria from one meal, then pass on the pathogens to whatever or whoever they feed upon next. Scientists believe nymphs infect people with Lyme disease more often than larval or adult ticks.
But given what Tsao and her collaborators observed, northern and southern ticks have different methods of climbing aboard. That variation in turn influences what sorts of animals they find as hosts. Lyme disease-causing bacteria are still circulating among ticks and animals in the South, of course, but are not infecting people quite so often as they do in the Midwest and Northeast.
“Even if they were highly infected, a person’s probability of running into one of these ticks just by walking through the woods is much lower in many areas of the South than it is in the North,” Tsao said.
Regional differences in tick genetics and behavior also collide with the timing of the tick life cycle — when adults that have huddled up for winter reemerge, when larvae are hatching, when nymphs are feeding, and coming full circle, when adults are laying eggs — which can vary with regional weather patterns.
The American dog tick (Dermacentor variabilis), also known as the wood tick, is larger than the black-legged tick (Ixodes scapularis), which is also called the deer tick.

Armed Forces Pest Management Board (CC BY-NC-ND 2.0)
“The larvae get infected by feeding on an infected animal, usually in the summer,” explained Columbia University entomology professor Maria Diuk-Wasser via email. “They then molt (grow) to nymphs and spend the winter in ‘hibernation.’ The following year, they emerge early to infect the next generation of mice, which then infect then next generation of larvae, and so on. If the timing of feeding (nymphs before larvae) gets disrupted, transmission of the bacterium stops. This seems to happen in some areas in the South.”
One caveat in these regional differences is that just like the Midwest, the South encompasses a wide variety of habitats.
“Inland states like Kentucky, Tennessee and Alabama have large forest areas that are hot and dry, with leached clay soils — none of which makes for good black-legged tick habitat,” said Graham Hickling, a professor of forestry, wildlife, and fisheries at the University of Tennessee.
“Black-legged ticks are still found in such areas, but generally at very low densities. In contrast, in humid, sandy habitats like coastal Florida, and in some major river valleys in inland states, black-legged tick densities can be high and similar to those seen in northern Lyme-endemic states.”
Different strokes, different hosts
Those ground-favoring ticks in the South really like to feed on reptiles, including the southeastern five-lined skink (Plestiodon inexpectatus) and the broad-headed skink (Plestiodon laticeps). Reptiles are thought to be less capable of harboring Lyme disease-causing bacteria than other animals ticks feed upon, namely birds and mammals.
In the western U.S., for instance, a protein in the blood of the western fence lizard (Sceloporus occidentalis) effectively clears out the bacteria from ticks that feed on it. While both northern and southern ticks also often feed on rodents, northern ones are much more likely to feed on white-footed mice, which have a proven record of harboring Lyme pathogens.
“The ones in the South evolved around a different set of vertebrate hosts than the ones in the North,” said Charles Apperson, a professor emeritus of entomology at North Carolina State University. “The ones in the North evolved over time where there are plenty of small rodent hosts that we now know are the source of the [Lyme disease] spirochete.”
Apperson and other Lyme disease researchers are mostly dealing with the spirochete Borrelia burgdorferi, the species that is primarily responsible for causing Lyme disease across the U.S. Scientists discovered another North American Lyme-causing bacteria, Borrelia mayonii, in 2013, and it has been found in patients from Wisconsin, Minnesota and North Dakota. In Europe, Borrelia garinii and Borrelia afzelii are the most common culprits.

Tony Alter (CC BY 2.0)
The southeastern five-lined skink is a more common meal for black-legged ticks in the southern U.S. than it is in more northern climes.
The mystery is that lizards can harbor Lyme disease bacteria. In Europe and northern Africa, research has shown that some lizard species can be an effective reservoir for Borrelia lusitaniae, another bacteria species that causes Lyme disease.
Apperson has conducted experiments in which five-lined skinks and green anoles (Anolis carolinensis), both abundant in the southeastern U.S., were injected with Borrelia burgdorferi. Tick larvae then fed on the lizards. Of the 424 ticks that fed on infected skinks, 100 came away infected themselves. Only 47 ticks fed on the green anoles, though, only one was infected, suggesting the skinks were more capable than the anoles of passing on the bacteria to ticks.
However, as Apperson admits, this experiment doesn’t say a whole lot about how much of a vector these lizards might be in real-life Lyme-related ecology. The test subjects were confined in a lab setting and therefore under stress, and the scientists gave them a “very high dose” of Borrelia burgdorferi. When an animal is under such conditions, all sorts of things can go out of whack, including its immune responses.
“How that relates to nature, I don’t know,” Apperson said, but added that it does demonstrate that lizards can help Lyme disease spread under the right circumstances.
To further complicate these questions, even host animals that don’t readily harbor Lyme disease can affect its spread.
In Wisconsin, Ixodes scapularis tends to feed a lot on deer, hence one of its colloquial names. But scientists studying the ticks’ range of animal meals have observed that deer don’t seem to be very good reservoirs for Borrelia burgdorferi. Additionally, ticks feeding on deer tend to be adults, which means they’re taking their last blood meal and won’t go on to potentially infect another host with any pathogens they might be carrying. Still, female adults go on to lay eggs after that meal, so the fortunes of deer can still indirectly impact tick reproduction and the next generation’s ability to spread Lyme disease.

Graham Hickling/University of Tennessee
Female (above) and male (below) black-legged ticks feed on a deer, with only the former becoming engorged with a blood meal
Entomologists and disease ecologists believe that other mammal hosts, chiefly the white-footed mouse (Peromyscus leucopus), are primarily responsible for passing Lyme-causing bacteria on to ticks. White-footed mice are so important to this field of study that a forest floor dense with acorns is considered a terrifying signal of an impending tick infestation. Mice will come out in droves to eat those acorns, and the ticks will be waiting in the woods to gorge on mouse blood and acquire the bacteria.
The white-footed mouse is a common carrier of the bacteria that causes Lyme disease.

Wisconsin Department of Natural Resources (CC BY-ND 2.0)
Wisconsin has its share of reptiles that can serve as hosts for ticks. Researchers working at Fort McCoy near Sparta, as part of a multi-university project Michigan State ecologist Jean Tsao leads, have collected deer ticks from five-lined skinks and snakes.
But reptiles are simply not as prominent in the state as Borrelia-spreading rodents. The five-lined skink’s range in Wisconsin includes a roughly Y-shaped swath of the state, which doesn’t include the northwestern counties that tend to see the highest Lyme infection rates. (White-footed mice are found just about all over the state.) And Tsao said that in her experience, it’s very rare for researchers to find ticks on a snake in either the Midwest, the Northeast or the South.
Climate change and ticks on the move
So much about Lyme disease hinges on weather, which could also help explain regional variations.
Howard Ginsberg, a Rhode Island-based research ecologist with the U.S. Geological Survey, released a study in 2017 that explored how heat and humidity might relate to regional variations in Lyme disease infection rates. Large swaths of the continental U.S. have high humidity during the summer, but the southern part of the nation is generally hotter than the Northeast or Upper Midwest. Ginsberg’s experiments found that tick larvae generally lived longer in “northern” temperatures (slightly lower temperatures, if still hot) than in “southern” ones when relative humidity was low for both. When humidity was moderate or high, that gap between northern and southern survival rates closed.
These findings might mean that while humidity is good for ticks regardless of region or temperature, ticks in hotter areas will come under more stress when humidity flags. It might have something to do with the lower rates of Lyme disease in the southern U.S.
Ginsberg is still trying to figure out exactly what the connection is — it may be that heat drives southern ticks to spend more time hiding out under leaves on the ground, or that the heat makes it easier for these ticks to dry up and die, or perhaps both. Then there’s the larger question of how temperature and humidity conditions interact with all the other factors helping and hurting ticks.

Centers for Disease Control and Prevention
Climate change could also upend the geography of tick-borne diseases. Warming temperatures are helping ticks spread their geographic range, and fewer days below freezing means more days to crawl around, feed, and ultimately, spread pathogens. The lone star tick (Amblyomma americanum) is expanding northward, including into Wisconsin, bringing with them their own brew of pathogens, including a bizarre meat allergy.
Wisconsin has also experienced movement in another direction; deer ticks, still mostly concentrated in the northern and western reaches of the state, are spreading towards the south and east.
Meanwhile, northern ticks might be moving southwards, and with them the specific strains of bacteria they carry.
“In states like Tennessee, we are starting to find tick populations infected with a mixture of northeastern and Midwestern strains of Borrelia,” said Graham Hickling of the University of Tennessee.
North Carolina State’s Charles Apperson said the northeastern branch of the Ixodes scapularis population is expanding southward via the Appalachian Mountains, but not so much along the Atlantic Coast.
This pattern would seem to match up with Hickling’s observation that tick populations are “surging” in places like Tennessee, Kentucky, and inland areas of Virginia and North Carolina. But Hickling noted this could be explained by a boom in the existing southern tick population, perhaps mixed with some migration from the north.
“If northern ticks are spreading south, we would expect to see bites from black-legged ticks, and locally-acquired Lyme disease cases, to shift increasingly into the summer months, since adult black-legged ticks are winter- and spring-active,” he said. “I haven’t seen any good evidence for this, so far, in part because Lyme case reporting from southern states doesn’t usually separate out travel cases from the North vs locally-acquired cases.”
Hickling hopes to work with health officials around the South to develop a clearer understanding of that data.
Changing weather patterns around North America will also affect the timing of ticks’ life cycles and feeding habits, explained Columbia University entomologist Maria Diuk-Wasser, which may have just as much impact on Lyme disease as tick migrations.
In the South, one open question is what happens when northern and southern deer ticks intermingle. Will the resulting offspring’s questing behavior favor the influence of one lineage or another, and if so, which one?
“They may also be hybridizing — mating — with southern ticks, and we don’t know which behavior will predominate,” Diuk-Wasser said.
This report was produced in a partnership between Wisconsin Public Radio, Wisconsin Public Television and the University of Wisconsin Cooperative Extension. © Copyright 2018, Board of Regents of the University of Wisconsin System and Wisconsin Educational Communications Board.
Series: Tick-Borne Diseases In Wisconsin: https://www.wiscontext.org/tick-borne-diseases-wisconsin
______________
**Comment**
Please know this idea of climate change affecting ticks and therefore transmission of disease to humans is contested by tick experts: https://madisonarealymesupportgroup.com/2017/08/14/canadian-tick-expert-climate-change-is-not-behind-lyme-disease/
The reason this is important is it takes the focus off of what is more important – very sick people and their need for timely diagnosis and effective treatment.
It’s been 40 years and these issues haven’t changed, yet researchers are lining their pockets with lots of money for climate change research.
Please know every time you hear the words, “Climate change,” there is a cash register chinging in the background as it represents huge research dollars but does absolutely nothing for patients.
According to John Scott, an independent researcher who’s infected with Lyme himself, ticks are marvelous ecoadaptors and will survive anything.
In the end, it’s about very sick people who need answers so they can get better. Never forget that.








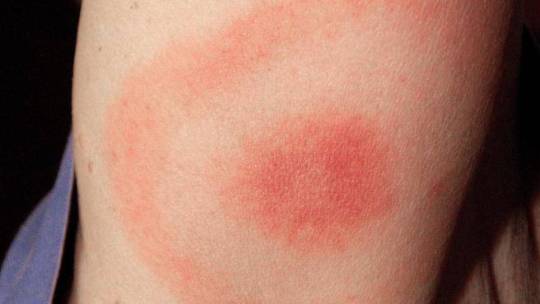 The bullseye rash, like the one pictured on a woman’s arm above, is often associated with the early stages of Lyme disease. (JAMES GATHANY)
The bullseye rash, like the one pictured on a woman’s arm above, is often associated with the early stages of Lyme disease. (JAMES GATHANY)