Live Q & A Webinar: Lyme Coinfections 11/19
|
|||||
|
|||||
|

|
|||||
|
|||||
|
Category:
Activism, Babesia, Bartonella, Candida, Lyme, Mycoplasma, Viruses
https://www.galaxydx.com/difference-between-bartonella-and-lyme-disease/
written on
Bartonella and Lyme Borrelia are easily confused because they are both vector-borne bacteria with similar sounding names and frequently have complex, interrelated roles in chronic illnesses. Patients often believe Bartonella and Borrelia are the same before speaking to us, or they may mistake something they read online about one for the other. In this post, we are going to describe some unique characteristics that distinguish these bacteria from each other.
The morphology of bacterial cells is based on their typical size, shape, and arrangement. These attributes may be different across various settings, such as in culture or patient samples. These differences may seem trivial to someone who is not a biochemist, but they can have profound effects at the molecular level. For example, proteins located on the cell wall and membrane are often targets for diagnostic assays and medications. Depending on morphological characteristics, these surface proteins may be less accessible or have entirely different chemical makeups. This is one reason there are so many types of antibiotics.
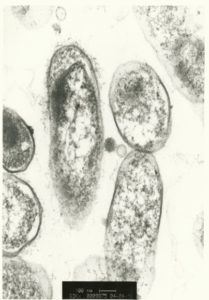
There are almost 40 characterized species of Bartonella about 15 of which are implicated in human illnesses. They are gram-negative, rod-shaped bacteria that grow very slowly. Their average dividing time is about 22-24 hours, which is much longer than the 20 minutes that E. coli species (another rod-shaped bacteria) require. Their average size is about 2 micrometers in length and 0.5 micrometers in width. One unique characteristic of Bartonella is that their membrane has a “Type IV Secretion System” that supports their ability to attach to and invade various target cells, such as endothelial and red blood cells.
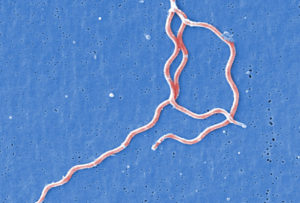
There are more than 50 named Borrelia species, with 3 primarily being implicated in human illnesses. They are like Bartonella in that they take a long time to divide (~12-18 hours) and stain weakly like gram-negative bacteria. However, unlike Bartonella, they are considered spirochetes due to their unique spiral shape and highly specialized cell walls and membranes. The primary species that causes Lyme disease in North America is Borrelia burgdorferi. This species has unique surface proteins called OspA, OspB, and OspC that are important for pathogenicity.
Bartonella and Borrelia species are both vector-borne bacteria, meaning they can be transmitted between hosts via a “competent” vector. A competent vector is typically an arthropod, like a tick or flea, that can acquire the pathogen during a meal and then transmit viable bacteria to another host in a subsequent feeding or through its excrement (feces). Bartonella and Borrelia both require access to the bloodstream for infection to occur, but research shows that Bartonella species have quite a few more options for getting there compared to Borrelia. Below is a list of confirmed as well as suspected vectors of each.
Bartonella and Borrelia species both cause acute and chronic infections that can be difficult for physicians to discern unless the patient has known exposure and recognizable clinical symptoms. Unfortunately, the latter is not the reality for many patients who become infected. Patients may first be diagnosed with another condition, like lupus or multiple sclerosis, before having testing done for bartonellosis or Lyme borreliosis. Why do these very different illnesses have similar symptoms?
The simple answer is that the body has a limited number of ways that it can respond to stress from bacterial infections. This host response is the result of a combination of differences between people in general health, genetic factors, and immune status that are integral to determining how an individual will respond to an infection. One person may experience neurologic symptoms, whereas another might only experience joint pain despite having the same infection. This makes it extremely difficult to define clinical patterns for infections, especially those that can lead to multi-systemic disease like bartonellosis and Lyme borreliosis.
Differentiating Symptoms
The main differences between bartonellosis and Lyme borreliosis symptoms typically occur early in the infection. However, many patients do not experience the “classic” signs that define the acute infections.
Cat scratch disease (Bartonella henselae infection) is described as fever and lymph node swelling near the location of a cat scratch or bite. However, this acute presentation of bartonellosis is not the only course of infection. Trench fever (Bartonella quintana infection) and Carrion’s disease (Bartonella bacilliformis infection) come with their own set of symptoms.
Bartonellosis may be associated with long, thin, red marks on the skin called striae, but more research is required to understand Bartonella species’ role. However, it is associated with a skin condition called bacillary angiomatosis primarily in immunocompromised individuals.
The CDC reports that 20-30% of patients who are exposed to Borrelia burgdorferi develop a bullseye rash called erythema migrans. This rash can also have other appearances. Most patients do not have the rash, or they may not see the rash if it is small or covered by hair. They can also experience facial palsy or extremely swollen joints.
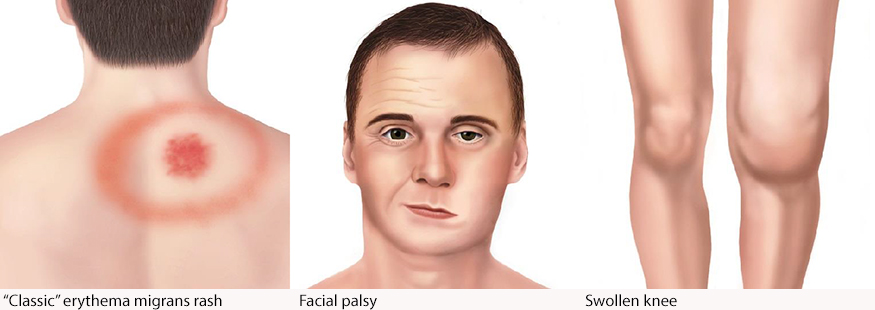
To find out more about these skin symptoms, see our blog post “Vector-borne Disease and Skin: Your First Layer of Defense.”
The symptoms of bartonellosis and Lyme borreliosis are similar because what the patient feels in everyday life is largely mediated by the host response. However, key differences between their causative pathogens make them each a disease of their own with their own unique challenges for testing and treatment.
Deng, H. et al. (2018). Molecular mechanisms of Bartonella and mammalian erythrocyte interactions: A review. Frontiers in Cellular and Infection Microbiology, 8, 431. doi:10.3389/fcimb.2018.00431 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6299047/
Breitschwerdt, E. B. (2017). Bartonellosis, One Health and all creatures great and small. Veterinary Dermatology, 28(1), 96-e21. doi:10.1111/vde.12413 https://www.ncbi.nlm.nih.gov/pubmed/28133871
Cheslock, M. A., & Embers, M. E. (2019). Human bartonellosis: An underappreciated public health problem? Tropical Medicine and Infectious Disease, 4(2), 69. doi:10.3390/tropicalmed4020069 https://www.mdpi.com/2414-6366/4/2/69/htm
Centers for Disease Control and Prevention. (2015). Louse-borne relapsing fever (LBRF). Available at https://www.cdc.gov/relapsing-fever/resources/louse.html
__________________
For more: https://madisonarealymesupportgroup.com/category/bartonella-treatment/
https://madisonarealymesupportgroup.com/category/lyme-disease-treatment/
Also, please know that borrelia has been found in mosquitoes. While transmission hasn’t been proven conclusively, many patients swear they became ill after a suspicious mosquito bite: https://sponauglewellness.com/lyme-study-how-borrelia-bacteria-is-transmitted-from-mosquitoes-to-humans/ In response to the time needed to transmit, recently this came out on how ticks can spread Rickettsia immediately: https://madisonarealymesupportgroup.com/2019/11/14/study-shows-ticks-can-transmit-rickettsia-immediately/ Borrelia can also be in the salivary glands of ticks which also could make transmission happen much more quickly than the 24-48 hours we’ve been told it takes.
Also, the elephant in the room is that Lyme and Bartonella both can possibly be transmitted in other ways: https://madisonarealymesupportgroup.com/2019/06/20/lyme-disease-transmission-can-it-spread-from-person-to-person/ Very balanced article but make sure and read my comments after the article. I include links to a PhD who is an expert on borrelia and she’s isolated it from mosquitos, fleas, mites, semen, urine, blood, spinal fluid, and tears, indicating the potential to be spread on hands person to person.
Remember – everyone has a bias whether they admit to it or not.
Category:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6812694/
Bartonella henselae is a relatively uncommon pathogen that can present as a serious disease in immunocompromised patients. We present a case of a 76-year-old man with stable chronic lymphocytic leukemia (CLL) who presented to the emergency department (ED) with an onset of right axillary lymphadenitis after recovering from a recent cat bite on the ipsilateral finger. Doppler ultrasound demonstrated an irregular, circumscribed 5cm x 4cm, hypoechoic mass with mild vascular flow consistent with an enlarged abnormal lymph node. The patient was diagnosed with cat scratch disease and discharged on oral antibiotics with spontaneous drainage of the purulent materials in subsequent outpatient oncology visits. This case highlights the classic presentation of this rare disease in an immunocompromised patient with feline contact. Early antibiotics should be considered for at-risk and immunocompromised patients due to low sensitivity and specificity for Bartonella serologic tests. CLL can also present with similar progressive lymphadenopathy with severe systemic symptoms and extranodal involvement that requires emergent oncologic interventions and diagnostic vigilance.
________________
**Comment**
Bartonella is NOT rare.
In this article, Dr. Mozayeni talks about Bartonella being one of the major co-infections of Lyme disease. It’s even more prevalent than Lyme, as there are many more ways to contract the disease (eg. flees, cats). In a study, that Dr. Breitschwerdt and Mozayeni published in The Journal of Emerging Diseases, about 60% of Lyme patients tested positive for Bartonella: https://madisonarealymesupportgroup.com/2016/01/03/bartonella-treatment/ If you want to know the likelihood of infection, please see the checklist within the article. Print it out and check how many symptoms you have. Take this with you to your doctor appointment and discuss the likelihood.
I’m glad this research article points out that testing is poor. This is true of each and every test for tick borne illness. This requires an educated and experienced practitioner to discover what patients are infected with. If you have tick borne illness, save your time and money and get to an ILADS trained doctor.
Category:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6804791/
To describe a patient whose retinal findings suggestive of tick-borne disease but evaluations led to early diagnosis and treatment of human immunodeficiency virus (HIV) infection.
A young patient presented with bilateral uveitis, branch retinal artery occlusion and retinal findings suggestive of infective/inflammatory etiology. Laboratory evaluations revealed that the patient was positive for co-infection with Rickettsia conorii and Bartonella henselae. On further investigation, the patient tested positive for HIV infection. The patient was treated with doxycycline as well as highly active anti-retroviral therapy (HAART) to control both opportunistic infections as well as HIV infection.
Patients with HIV infection are at risk for multiple, simultaneous opportunistic co-infections, including those with tick-borne diseases.
________________
**Comment**
A great example of how infections can drive viruses. Mainstream medicine is still hopelessly in the dark on the seriousness of tick borne infections and the polymicrobial nature of them: https://madisonarealymesupportgroup.com/2018/10/30/study-shows-lyme-msids-patients-infected-with-many-pathogens-and-explains-why-we-are-so-sick/
Category:
October 16, 2019
This next case study is of an 18-year-old female who was adopted at the age of 5. Her adoptive mother described her as a malnourished premature baby who eventually received good foster care. This young lady was diagnosed with a growth hormone deficiency that was left untreated in her country of origin at the age of two. By the age of five, she was adopted and moved to the US with her American family. She was fully immunized twice, diagnosed with hypothyroidism and inadequate growth. By this time, an Endocrinologist was onboard and treating her thyroid and growth deficiencies. She seemed to rebound, reaching puberty by the age of 13. Life was stable for some time until January of 2016. She was nearly sixteen years old and developed sudden neuropsychiatric symptoms with acute confusion, severe obsessive-compulsive disorder, frequent urination, insomnia, auditory hallucinations, severe sensory issues, leg tremors and eventually catatonia. Given her acute changes, her mother rushed her to the Emergency Room for evaluation. EEG was negative and she was hospitalized for apparent acute psychosis treated with Risperdal and Ativan.
After her hospitalization she followed up with a well-known Neurologist who identified positive Mycoplasma and initiated a course of Azithromycin. By the fourth dose she began to return to her normal state and began sleeping again. She was treated for over a month with antibiotics and seemed stable.
There was a great deal of stress in the family, a close family member died and within two weeks she developed new onset grand mal seizures while sleeping. Another ER visit with a normal EEG at the time determined perhaps the stress and trauma of her family member’s death may have triggered the event.
In January 2018 she had another grand mal seizure early in the am. Her neurologist began medications to address. She had no additional seizure activity but noted increasing anxiety. By December 2018 she suffered another grand mal seizure.
Further evaluation by the neurologist showed negative Lyme screening only, viral panels negative, tick-borne co-infections were not obtained, thyroid studies, electrolytes and inflammatory markers were all within normal limits.
This patient presented to me in February 2019. Upon further evaluation she was found to have progressive muscle weakness, cognitive dysfunction ongoing psychiatric symptoms, tremors and noted random striae or “stretch-marks” that would appear and disappear all over her body. She stated that this had occurred since the age of fourteen. She admitted several evaluations with psychiatric acute hospital admission for escalating neuropsychological symptoms that included visual and auditory hallucinations, compulsions, rage, emotional lability, delusions, anxiety as well as the ongoing physical symptoms. Neuropsychological meds were ineffective. The patient upon presentation was taking high dose Depakote, gabapentin and folic acid to control her seizure activity.
Initial lab work up at my office showed an IGM positive Bartonella Henselae, Lyme Western Blot with an IGM indeterminate band 23-25 and IGG positive bands 18,23-25,28,31,34,39,41,45,and indeterminate bands 58 and 66. She also showed IGG positive Rickettsia and Anaplasma. She carried one copy of MTHFR A1298C and had significant GI bacteria overgrowth with Streptococcus, Citrobacter, Proteus and Bacillus.
She was started on a course of Azithromycin and Bactrim as well as biofilm busters and herbals. Two months later she reported significant improvements noting striae lightening, energy improvements, mood stability, resolution of hallucinations, and her sleep was improving. She noted ongoing body and hand tremors as well as struggles cognitively with word finding but was back in school full time.
We decided to continue the treatment course and repeat her bloodwork in two months as well as continue follow up with her Neurologist to monitor. By June the patient was feeling great. She began a Depakote wean with her Neurologist and graduated High School.
Her lab results showed improvements with Bartonella levels as well as GI bacterial overgrowth. Rickettsia antibodies lingered unchanged as did Lyme bands. I added to her regimen Doxycycline and Cefdinir as well as an antifungal and supportive herbals to prevent yeast.
This patient is still a work in progress, however what is important to note is her complete reversal of the neuropsychological symptoms once antibiotics were initiated as well as the ongoing, successful wean of seizure medications.
Bartonella and Rickettsia infections both have an affinity for the central nervous system. It is challenging to identify given their non-specific symptom presentation at times. Rickettsia isn’t well understood regarding brain parenchyma and central nervous system transmission. We know in mouse studies, Rickettsia and Bartonella both contribute to neuroinflammation which can contribute to acute psychological symptoms. We see this type of neurological process in classic PANS patients related to strep. Although I see the trend clinically, I don’t feel that autoimmune encephalopathy related to tick-borne infections in children and young adults is well documented.
My hope is thru case study presentations you’ll connect real world, everyday struggles of these vulnerable patients with the disease process. I strongly feel further exploration of autoimmune encephalopathy as it relates to Lyme and other Tickborne illnesses in pediatrics should be a collaborative effort with mental health practitioners and welcome those interested to contact me.
__________________
For more: https://madisonarealymesupportgroup.com/2016/01/03/bartonella-treatment/
https://madisonarealymesupportgroup.com/2016/02/07/mycoplasma-treatment/
https://madisonarealymesupportgroup.com/2016/03/08/anaplasmosis/ (Treatment)
https://reference.medscape.com/article/968385-treatment (Rickettsia treatment)
Category:
Bartonella, Inflammation, Lyme, Mycoplasma, Psychological Aspects, research, Treatment