https://thevaccinereaction.org/2020/09/coronavirus-cases-plummet-when-pcr-tests-are-adjusted/
By Barbara Caceres
Published September 29, 2020 | Medicine, Public Health
Health experts now say that PCR testing for SARS-CoV-2, the virus associated with the illness COVID-19, is too sensitive and needs to be adjusted to rule out people who have insignificant amounts of the virus in their system.1 The test’s threshold is so high that it detects people with the live virus as well as those with a few genetic fragments left over from a past infection that no longer poses a risk. It’s like finding a hair in a room after a person left it, says Michael Mina, MD, an epidemiologist at the Harvard T.H. Chan School of Public Health.2
In three sets of testing data that include cycle thresholds compiled by officials in Massachusetts, New York and Nevada, up to 90 percent of people testing positive carried barely any virus, a review by The New York Times found.3
Manufacturers and Labs Set Criteria for Positive COVID-19 Test Results
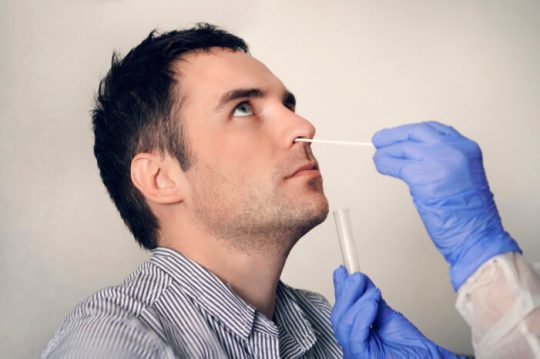
The reverse transcriptase quantitative polymerase chain reaction (RT-qPCR) test used to identify those people infected with the SARS-CoV-2 virus uses a nasal swab to collect RNA from deep within the nasal cavity of the individual being tested. The RNA is reverse transcribed into DNA and amplified through 40 or more cycles, or until virus is detected.4 The result is reported as a simple “yes” or “no” answer to the question of whether someone is infected.
The U.S. Food and Drug Administration (FDA) officials state they do not specify the cycle threshold ranges used to determine who is positive, and that commercial manufacturers and laboratories set their own threshold ranges.5
PCR Test Threshold for COVID-19 Positivity Is Too Sensitive
Any test with a cycle threshold (CT) above 35 is too sensitive, says Juliet Morrison, PhD, a virologist at the University of California, Riverside. “I’m shocked that people would think that 40 [cycles] could represent a positive.” A more reasonable cutoff would be 30 to 35, she added. Dr. Mina said he would set the figure at 30, or even less. Those changes would mean the amount of genetic material in a patient’s sample would have to be 100-fold to 1,000-fold that of the current standard for the test to return a positive result worth acting on.6
The CDC’s own calculations suggest that it is extremely difficult to detect any live virus in a sample above a threshold of 33 cycles.7
“We’ve been using one type of data for everything, and that is just plus or minus—that’s all,” Dr. Mina said. “We’re using that for clinical diagnostics, for public health, for policy decision-making.” But “yes” or “no” isn’t good enough, he added. It’s the amount of virus that should dictate the infected patient’s next steps. “It’s really irresponsible, I think, to forgo the recognition that this is a quantitative issue,” Dr. Mina said.8
The number of people with positive results who aren’t infectious is particularly concerning, said Scott Becker, executive director of the Association of Public Health Laboratories. “That worries me a lot, just because it’s so high,” he said.9
SARS-CoV-2 Positive Case Numbers Drop When Cycle Threshold is Adjusted, Removing Need for Contact Tracing
Officials at the Wadsworth Center, New York’s state lab, have access to CT values from tests they have processed, and analyzed their numbers at The Times’s request. In July, the lab identified 872 positive tests, based on a threshold of 40 cycles. With a cutoff of 35 cycles, about 43 percent of those tests would no longer qualify as positive. About 63 percent would no longer be judged positive if the cycles were limited to 30.
In Massachusetts, from 85 to 90 percent of people who tested positive in July with a cycle threshold of 40 would have been deemed negative if the threshold were 30 cycles, Dr. Mina said. “I would say that none of those people should be contact-traced, not one,” he said.
“I’m really shocked that it could be that high—the proportion of people with high CT value results,” said Ashish Jha, MD, director of the Harvard Global Health Institute. “Boy, does it really change the way we need to be thinking about testing.”10
“Gold Standard” PCR Tests Leave Many Unanswered Questions Due to Knowledge Gaps
A positive PCR test does not tell doctors whether the person is currently ill or will become ill in the future, whether they are infectious or will become infectious, whether they are recovered or recovering from COVID, or whether the PCR test identified a viral fragment from another coronavirus infection in the past. The CDC reports that a person who has recovered from COVID-19 may have low levels of virus in their bodies for up to three months after diagnosis and may test positive, even though they are not spreading COVID-19.11
CT Value Adds Context to PCR Results, Personalizes Care
Although the cycle threshold (CT) is not reported on PCR tests, new evidence suggests the CT value could help to better inform clinical decisions, particularly when testing in the absence of symptoms for COVID-19. When SARS-CoV-2 virus is detected after fewer amplification cycles, that indicates a higher viral load and a higher likelihood of being contagious, while virus detected after more amplifications indicates a lower viral load.
“It’s just kind of mind-blowing to me that people are not recording the CT values from all these tests—that they’re just returning a positive or a negative,” said Angela Rasmussen, PhD, a virologist at Columbia University in New York. “It would be useful information to know if somebody’s positive, whether they have a high viral load or a low viral load,” she added.12
In a study published in Clinical Infectious Diseases in May, 2020,13 the authors suggested that viral load based on CT cutoff could establish whether inpatients have transmissible disease or need to be retested. This would conserve valuable testing capacity, reagents, and personal protective equipment (PPE), and determine when a patient could discontinue isolation. Taking the CT value into account may also help justify symptom-based strategies recommended by the CDC. CT values may enable contact tracers to focus only on persons most likely to be infectious, which will become increasingly important as asymptomatic screening expands.
Another study14 found that patients with positive PCR tests at a CT above 33-34 are not contagious and can be discharged from the hospital or strict confinement at home.
Evidence from both viral isolation and contact tracing studies supports a short, early period of transmissibility. By accounting for the CT value in context, RT-qPCR results can be used in a way that is personalized, highly sensitive, and more specific.15
FDA Approves Rapid, Less Sensitive Coronavirus Antigen Test
Highly sensitive PCR tests seemed like the best option for tracking the coronavirus at the start of the pandemic. But for the outbreaks raging now, Dr. Mina said, what’s needed are coronavirus tests that are fast, cheap and abundant enough to frequently test everyone who needs it—even if the tests are less sensitive. “It might not catch every last one of the transmitting people, but it sure will catch the most transmissible people, including the super spreaders.”
The FDA noted that people may have a low viral load when they are newly infected. A test with less sensitivity would miss these infections. That problem is easily solved, Dr. Mina said: “Test them again, six hours later or 15 hours later or whatever,” he said. A rapid test would find these patients quickly, even if it were less sensitive, because their viral loads would quickly rise. People infected with the virus are most infectious from a day or two before symptoms appear till about five days after. But at the current testing rates, “you’re not going to be doing it frequently enough to have any chance of really capturing somebody in that window,” Dr. Mina added.16
When a patient is tested for the coronavirus, doctors typically tell them to stay home until the results come in. If a patient tests positive and faces a two-week quarantine, that means they could spend a total of three weeks in isolation. That’s a long time for anybody who has bills to pay or kids to care for, and it’s understandable that some people will continue working until the results come in. The problem is that anybody who does this with a serious infection is putting others at risk.17 Rapid tests can be helpful in these situations.
In late August, the U.S. Food and Drug Administration (FDA) approved the first rapid coronavirus test that doesn’t need any special computer equipment. Made by Abbot Laboratories, the 15-minute test will sell for U.S. $5 but still requires a nasal swab to be taken by a health worker.18 The Abbot test is the fourth rapid point-of-care test that looks for the presence of antigens rather than the virus’s genetic code as the PCR molecular tests do. 19
References: Angela Rasmussen, Ashish Jha, Association of Public Health Laboratories, Barbara Cáceres, bbot Laboratories, CDC, Centers for Disease Control and Prevention, Clinical Infectious Diseases, Columbia University, coronavirus, COVID-19, FDA, Food and Drug Administration, Harvard Global Health Institute, Harvard T.H Chan School of Public Health, Juliet Morrison, National Vaccine Information Center, NVIC, PCR tests, personal protective equipment, PPE, reverse transcriptase quantitative polymerase chain reaction, RT-qPCR, SARS-CoV-2, The Vaccine Reaction, Wadsworth Center
_____________________
**Comment**
We have precisely the opposite problem with Lyme/MSIDS testing. The arbitrary antibody cut off has kept the sickest from ever testing positive. This is another perfect example of how the tests are rigged for the CDC’s predetermined outcome. In the case of COVID, they want to show high numbers to buttress their fear-mongering. In the case of Lyme/MSIDS they want low numbers so everyone thinks it’s ‘rare’ and unimportant, and undeserving of research dollars.
If ‘authorities’ show sudden interest in Lyme/MSIDS it’s because there’s a vaccine in the pipeline.
And yes, Lyme testing is rigged: https://madisonarealymesupportgroup.com/2020/02/10/the-bitter-feud-over-lymerix/
