Small Fiber Neuropathy & PTLDS
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0212222
Published: February 12, 2019 https://doi.org/10.1371/journal.pone.0212222
Abstract
Objectives
To examine whether post-treatment Lyme disease syndrome (PTLDS) defined by fatigue, cognitive complaints and widespread pain following the treatment of Lyme disease is associated with small fiber neuropathy (SFN) manifesting as autonomic and sensory dysfunction.
Methods
This single center, retrospective study evaluated subjects with PTLDS. Skin biopsies for assessment of epidermal nerve fiber density (ENFD), sweat gland nerve fiber density (SGNFD) and functional autonomic testing (deep breathing, Valsalva maneuver and tilt test) were performed to assess SFN, severity of dysautonomia and cerebral blood flow abnormalities. Heart rate, end tidal CO2, blood pressure, and cerebral blood flow velocity (CBFv) from middle cerebral artery using transcranial Doppler were monitored.
Results
10 participants, 5/5 women/men, age 51.3 ± 14.7 years, BMI 27.6 ± 7.3 were analyzed. All participants were positive for Lyme infection by CDC criteria. At least one skin biopsy was abnormal in all ten participants. Abnormal ENFD was found in 9 participants, abnormal SGNFD in 5 participants, and both abnormal ENFD and SGNFD were detected in 4 participants. Parasympathetic failure was found in 7 participants and mild or moderate sympathetic adrenergic failure in all participants. Abnormal total CBFv score was found in all ten participants. Low orthostatic CBFv was found in 7 participants, three additional participants had abnormally reduced supine CBFv.
Conclusions
SFN appears to be associated with PTLDS and may be responsible for certain sensory symptoms. In addition, dysautonomia related to SFN and abnormal CBFv also seem to be linked to PTLDS. Reduced orthostatic CBFv can be associated with cerebral hypoperfusion and may lead to cognitive dysfunction. Autonomic failure detected in PTLDS is mild to moderate. SFN evaluation may be useful in PTLDS.
________________
**Comment**
Keep in mind, researchers by nature have to keep tight & narrow study parameters. In this case they chose to use the unscientific and abysmal CDC 2-tiered blood serology which research has shown misses over half of all cases. It was also never intended for diagnosis but for surveillance purposes only. Lyme should be a clinical diagnosis. We desperately need a better form of accurate testing for this very reason. Nearly ALL research is based upon 2-tiered serology which leaves out a huge subset of patients – which means there is study bias.
Some of the sickest people NEVER test positive using CDC criteria.
Also, please note:
All patients had received a course of antibiotics for Lyme disease including 3 weeks of oral doxycycline as recommended by Infectious Diseases Society of America (IDSA) guidelines [13,30]. Because of symptoms persistence, all participants were treated with additional antibiotics (data in S1 Table). The list may not be accurate and may underestimate the antibiotic therapy as many participants were treated in multiple institutions and as such medical records may be incomplete.
Many would argue that the entire premise of this research article is flawed in that the CDC moniker of PTLDS is not taking into account persistent/chronic infection. For a fantastic read on that: https://madisonarealymesupportgroup.com/2019/02/25/medical-stalemate-what-causes-continuing-symptoms-after-lyme-treatment/
Until this issue is resolved, people will be put into two camps based on a false premise, 1) Acute cases 2) PTLDS. There are numerous subsets of people. See link above.
Parasympathetic failure & low orthostatic CBFv was noted in 7 out of 10 patients and abnormal total CBFv score was found in all 10!
The parasympathetic system is responsible for stimulation of “rest-and-digest” or “feed and breed”[3] activities that occur when the body is at rest, especially after eating, including sexual arousal, salivation, lacrimation (tears), urination, digestion and defecation. https://en.wikipedia.org/wiki/Parasympathetic_nervous_system
Lyme loves the spine!
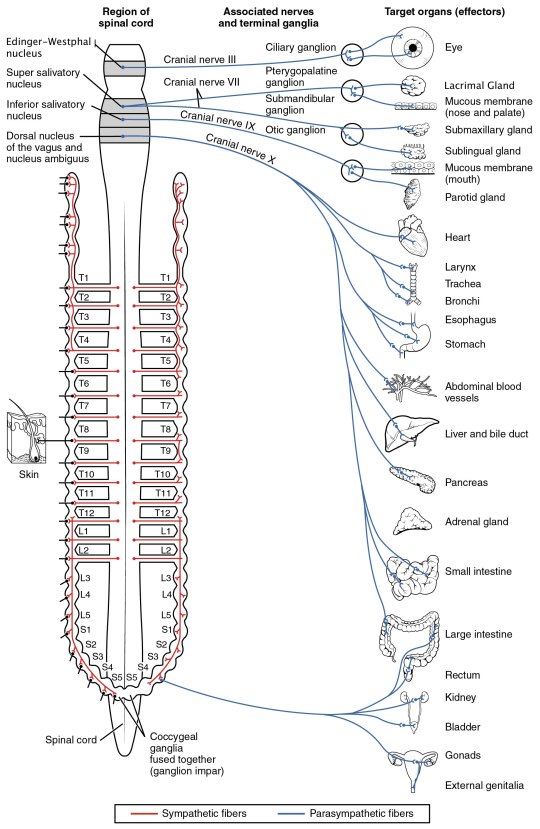
By OpenStax College – Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013., CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=30148020
You can visualize where on the spine the various organs receive information from. Notice all the things connected to the cranial nerves at the top. Please know Bartonella and other coinfections can affect these areas as well. Dr. Ericson’s found Bart all over the place: https://madisonarealymesupportgroup.com/2019/02/27/advanced-imaging-found-bartonella-around-pic-line/ Take a gander at her slides where she’s found Bart in collagen, where a PIC line was removed, skin biopsies (including the brain), cartilage, and blood cells.
Lastly, besides the spinal involvement, notice ALL of the patients had an abnormal Total CBFv score. This is cerebral blood flow velocity from the middle cerebral artery. Not enough blood flow = not enough oxygen, which is required for normal functioning. It alone can account for brain fog and fatigue. Too much blood flow = swelling and inflammation which can cause severe pain and even lead to Chiari: https://madisonarealymesupportgroup.com/2016/04/02/chiari/ In one week I met 3 Lyme patients with a Chiari diagnosis. I had an MRI to rule it out myself.
One drug that was most effective for me for this issue was Minocycline: https://madisonarealymesupportgroup.com/2017/06/04/minocycline-for-ms-and-much-more/

 1
1 