https://www.tickcheck.com/statistics?
Each tick submitted for testing contributes to the research being conducted at TickCheck. By keeping records of all the results generated, we have been able to gain valuable insights into disease prevalence and co-infection rates. The comprehensive testing panel has been especially helpful in contributing to this research by ensuring all diseases and coinfections are accounted for when examining a tick.
Our current research shows:
- 76% of ticks tested have at least one disease causing organism
- 49% are co-infected with two or more organisms
- 20% carry three or more
- 9% of the ticks tested carry four or more
Infection Visualization by Tick Species
All Ticks Tested
76% Positive for Infection
Negative (24%)
_____________________________
- 93% Positive for Infection
- Negative (7%)
- 63% Positive for Infection
- Negative (37%)
- 48% Positive for Infection
- Negative (52%)
Coinfection Visualization
- 2+ coinfection 49%
- No coinfection 51%
Pathogenic Prevalence
The information below shows the positive/negative prevalence ratio of selected pathogens we test for. These pathogens were observed in ticks from the United States and Canada. Data set includes tests performed since TickCheck’s founding in 2014 and is updated in real time. (
Go to link at beginning to filter by state. I’ve added the 3 listed for Wisconsin next to the entire sample size. Please note the small sample sizes of WI ticks.
Borrelia burgdorferi (deer tick) associated with Lyme disease
Sample size of 3,280 ticks. 70 Wisconsin ticks
- 30% postive 33% positive
- 70% negative 67% negative
Borrelia burgdorferi (western blacklegged tick) associated with Lyme disease
Sample size of 279 ticks.
Borrelia burgdorferi (lone star tick) associated with Lyme disease
Sample size of 899 ticks.
Borrelia burgdorferi (American dog tick) associated with Lyme disease
Sample size of 901 ticks.
Anaplasma phagocytophilum associated with anaplasmosis
Sample size of 2,146 ticks. 36 Wisconsin ticks
- 8% positive 11% positive in Wisconsin
- 92% negative 89% negative in Wisconsin
Babesia microti associated with babesiosis
Sample size of 1,894 ticks. 32 Wisconsin ticks
- 4% positive 6% positive
- 96% negative 94% negative
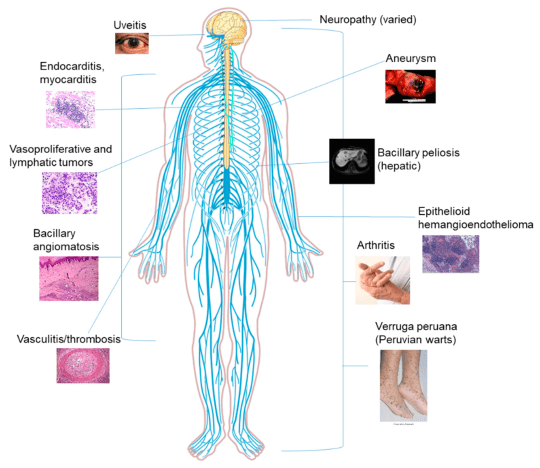
Bartonella spp. associated with bartonellosis
Sample size of 1,060 ticks.
- 47% positive
- 53% negative
Ehrlichia chaffeensis associated with ehrlichiosis
Sample size of 857 ticks.
Rickettsia spp. associated with Rocky Mountain spotted fever
Sample size of 944 ticks.
Francisella tularensis associated with tularemia
Sample size of 1,028 ticks.
Borrelia miyamotoi associated with B. miyamotoi
Sample size of 1,091 ticks.
Borrelia lonestari associated with STARI
Sample size of 831 ticks.
- 19% postitive
- 81% negative
Babesia spp. associated with babesiosis
Sample size of 564 ticks.
Mycoplasma spp. associated with Mycoplasma spp.
Sample size of 948 ticks.
Borrelia spp. associated with Borrelia spp.
Sample size of 612 ticks.
Powassan virus Lineage II associated with Deer tick virus
Sample size of 102 ticks.
- 24% positive
- 76% negative
Borrelia mayonii associated with Lyme disease
Sample size of 376 ticks.
Ehrlichia ewingii associated with ehrlichiosis
Sample size of 283 ticks.
Rickettsia amblyommii associated with Rocky Mountain spotted fever
Sample size of 177 ticks.
- 46% positive
- 54% negative
__________________
For more about Tickcheck: https://www.tickcheck.com/about
You can request free tick identification by sending in a quality picture of your tick. Using real-time PCR (Polymerase Chain Reaction), Tickcheck can determine the presence of certain pathogens with an accuracy level of over 99.9%. All information about how to send in your tick, costs of various tests, time for results, etc. is found here: https://www.tickcheck.com/info/faq
Jonathan Weber is the founder and CEO of TickCheck and became acutely aware of the dangers of tick-borne diseases after his father caught Lyme during a family trip on the Appalachian Trail.
___________________
**Comment**
This information supports current research showing many patients are infected with numerous pathogens causing more severe illness & requiring far more than the CDC’s mono therapy of doxycycline: https://madisonarealymesupportgroup.com/2018/10/30/study-shows-lyme-msids-patients-infected-with-many-pathogens-and-explains-why-we-are-so-sick/
It also supports previous work showing coinfections within ticks: https://madisonarealymesupportgroup.com/2017/05/01/co-infection-of-ticks-the-rule-rather-than-the-exception/
What I want to know is WHY nothing’s being done about this? Why are people STILL given 21 days of doxycycline when that particular med will not work on numerous pathogens?
Lastly, a word about statistics – this tick data should be used with caution & never to turn sick patients away due to a statistic. If you are the sorry sucker who gets bit by that ONE tick carrying a “statistically insignificant” pathogen, you still got bit and have to deal with it.
Shame on doctors for turning sick people away due to statistics and maps.
There’s no such thing as an “insignificant” tick bite!
But, Patients are STILL being turned away: https://madisonarealymesupportgroup.com/2019/04/22/its-just-crazy-why-is-lyme-disease-treatment-so-difficult-to-find-in-mississippi/
https://madisonarealymesupportgroup.com/2019/03/19/jacksonville-family-shares-daughters-9-month-diagnosis-of-rare-disease-which-isnt-rare-lyme/
https://madisonarealymesupportgroup.com/2018/05/31/no-lyme-in-the-south-guess-again/
https://madisonarealymesupportgroup.com/2017/10/24/no-lyme-in-oklahoma-yeah-right/
https://madisonarealymesupportgroup.com/2016/09/24/arkansas-kids-denied-lyme-treatment/ “They had the classic symptoms, they had the bulls eye rash, they had the joint pain, they had fevers and had flu like symptoms, yet we were denied treatment for at least two of them and I don’t understand how this is legal,” said Bowerman.
According to Dr. Naveen Patil, Director of the Infectious Disease Program, ADH,
“We don’t have Lyme Disease in Arkansas, we have the ticks that transmit Lyme Disease but we don’t have any recorded cases of Lyme Disease.”
Bowerman also received a letter from the clinic stating doctors would no longer treat her children because she consistently questioned their medical advice and recommendations.
This is getting to be way beyond ludicrous.