R Esfandyarpour, A Kashi, M Nemat-Gorgani, J Wilhelmy and RW Davis. 2019
The nanoelectric blood test

The research used nanomanufacturing techniques to embed large numbers of tiny electrodes within a silicon wafer. Each electrode, or nanoneedle, is comparable in size to a cell.
The researchers created a simplified blood sample for each patient that consisted of white blood cells (immune cells) in plasma, but without red blood cells and platelets. The scientists added each sample to a silicon wafer, and the electrodes then measured electrical impedance.
Impedance is a measure of how difficult it is for the electrical current to pass through the cells and/or plasma next to the electrodes. Critically, in the nanoelectric set-up, the authors say that change in impedance “results from cellular and molecular interactions”.
The key element of the test, which exposes the dramatic difference between patient and control samples, is to force the cells to use more energy than normal. The aim was to replicate at the cellular level a key aspect of ME/CFS: the way problems emerge when energy demands ramp up.
The researchers forced the cells to use more energy simply by adding sodium chloride — table salt — to the sample. In the jargon, the salt acts as a “hyperosmotic stressor”.
Some of the extra salt enters the cell and, through osmotic pressure, the salt draws water with it, causing the cell to swell up. The cell has to combat this tendency and so must use energy to power a molecular pump that pushes the extra salt back out of the cell.
The different response seen between the samples of patients and controls is striking. There is little change in electrical impedance for healthy cells. But after half an hour or so, there’s a huge increase in impedance for the samples from ME/CFS patients.
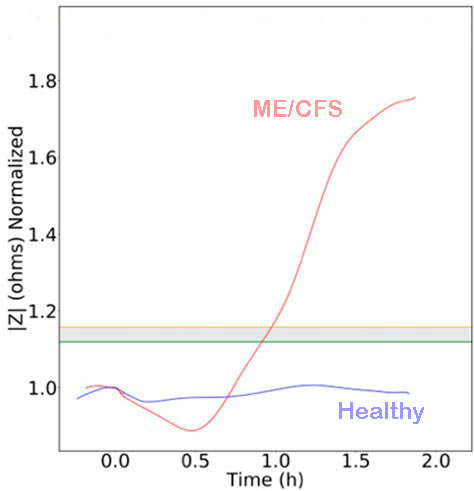
Lead author Rahmim Esfandyarpour told STAT News, “we’re forcing [patients’ cells to consume energy] and they are not happy… their reaction is different from the reaction of healthy cells”. The healthy cells seem to manage the situation comfortably.
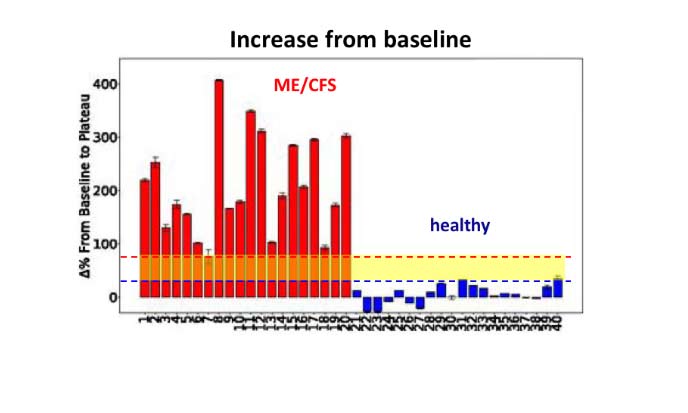
Strikingly, the increase in impedance for every single patient’s sample was substantially higher than for even the highest increase seen for any of the healthy controls’ samples.

It is the amount of clear daylight between patients and controls (indicated above by the yellow band in the graph) that makes these results so remarkable and so interesting. At the recent NIH ME/CFS conference, Dr Anthony Komaroff, a professor at Harvard Medical School, said that such a difference is “a clue to some underlying biology that could be causative of the symptoms of the illness.”
To make the findings more impressive, the authors also showed that the findings are highly reproducible. “If you test the same patient a week or month later, you get the exact same signal”, senior author Ron Davis told Medscape Medical News.
The study authors say that they believe these findings are unique to ME/CFS.
Commenting on the study, Chris Ponting, Professor of Medical Bioinformatics at Edinburgh University, said, “Excitingly, they appear to have discovered a distinguishing feature of ME/CFS, and one that can be measured simply and cheaply.” However, he stressed the need for replication of results and the need for sick controls. He said, “results should be replicated in a second cohort of individuals” and added that the device should be tested to see “whether it sets apart ME/CFS not just from general health but also from other disorders.”
Happily, the authors are planning to do just this. They have announced that they will be running a replication in a larger group of patients, — and will be including people with similar diseases as controls.
The researchers admit that they don’t know what biological differences lie behind the dramatic difference that the nanoelectricdevice shows between patients and controls. They speculate that they could be changes in the outer membrane of patients’ cells, amongst other possibilities. But the researchers are planning experiments to try to uncover the biology. The new work could be critical in understanding ME/CFS.
The authors are also working on adapting the technology to create a user-friendly platform for screening potential drugs. The basic idea is that any drugs that can make ME/CFS cells behave like healthy ones might be therapeutic in patients.
And the team have already started screening drugs that have already been approved for other conditions. If any of these prove effective in ME/CFS, they would be available to ME/CFS patients in a shorter timescale because they would have already passed through much of the regulatory process.
One of the most important uses for this new technology, if it proves to be accurate and if the differences are specific to ME/CFS, would be in helping to make robust diagnoses.
Davis’s team are already trying to adapt the technology so that it could be used in any doctor’s office (for now, it needs to be done in a lab). And the nanoelectric chips are very cheap to make commercially, so the test should be affordable and widely available.
In conjunction with existing measures, such as the Canadian Consensus Criteria, the nanoelectric device would make it relatively straightforward for physicians without specialist expertise to make a diagnosis.
Research in the US indicates that perhaps 80% of people with ME/CFS are undiagnosed. So there could be a million Americans who are sick with ME/CFS but don’t know what’s making them ill, and many more such people worldwide.
With a diagnosis, people could at least get advice on how to manage their condition more effectively until good treatments are available. That could improve life for a great many people, with the potential for making a huge difference once there are effective treatments.
___________________
For more: https://madisonarealymesupportgroup.com/2015/10/17/can-mecfs-be-caused-by-lyme/
https://madisonarealymesupportgroup.com/2019/03/12/unrest-documentary-about-me-cfs-on-netflix/
