Improved serodiagnostic performance for Lyme disease by use of two recombinant proteins in ELISA as compared to standardized two tier testing
Bradshaw GL, Thueson RK, Uriona TJ.
Journal of Clinical Microbiology, online first, 2017 Aug 2.
https://doi.org/10.1128/JCM.01004-17
Abstract
The most reliable test method for the serological confirmation of Lyme Disease (LD) is a 2-tiered method recommended by the CDC in 1995. The first-tier test is a low specificity ELISA test and the second-tier tests are higher specificity IgG and IgM Western blots. This study describes the selection of two Borrelia burgdorferi-recombinant proteins and evaluation of their performance in a simple 1-tier test for the serological confirmation of LD.
These two proteins were generated from
(a) the full length dbpA gene combined with the invariable region 6 of the VlsE gene (dbpA/C6) and
(b) the full length OspC gene.
The expressed dbpA/C6 and the OspC proteins were useful in detecting anti-Borrelia IgG and IgM antibodies, respectively. A blind study was conducted on a well-characterized panel of 279 human sera from the CDC comparing ELISA tests using these two recombinant antigens with the 2-tiered test method. The two methods (dbpA/C6-OspC vs 2-tiered) compared equivalently in identifying sera from negative control subjects (99% vs 100% specificity, respectively) and in detecting stage II & III LD patient sera (100% vs 100% sensitivity). However, the dbpA/C6-OspC ELISA test was markedly better (80% vs 63%) than the 2-tiered test method in detecting anti-Borrelia antibodies in stage I LD patients.
The finding suggest that these antigens could be used in a simple 1-tier ELISA assay that is faster to perform, easier to interpret, and less expensive than the 2-tiered test method, and which is better at detecting Borrelia specific antibodies in patient sera with stage I LD.
More on testing:
https://madisonarealymesupportgroup.com/2017/04/12/comparing-lyme-testing-with-hiv-testing/
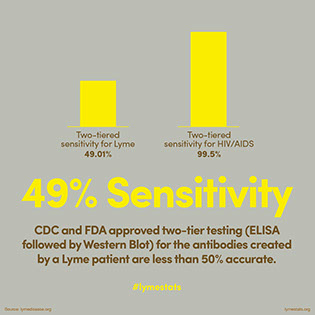


https://www.lymedisease.org/lyme-basics/lyme-disease/diagnosis/ Testing explained and interpreted.
