COVID-19 mRNA Shots Are Legally Not Vaccines & You’ll Never Guess What’s in the Tests
https://articles.mercola.com/sites/articles/archive/2021/02/09/coronavirus-mrna-vaccine
COVID-19 mRNA Shots Are Legally Not Vaccines
STORY AT-A-GLANCE
- By referring to COVID-19 vaccines as “vaccines” rather than gene therapies, the U.S. government is violating its 15 U.S. Code Section 41, which regulates deceptive practices in medical claims
- The mRNA injections are gene therapies that do not fulfill a single criteria or definition of a vaccine
- COVID-19 “vaccines” do not impart immunity or inhibit transmissibility of the disease. They only are designed to lessen your infection symptoms if or when you get infected. As such, these products do not meet the legal or medical definition of a vaccine
- Since a vast majority of people who test positive for SARS-CoV-2 have no symptoms at all, they’ve not even been able to establish a causal link between the virus and the clinical disease
- By calling this experimental gene therapy technology a “vaccine,” they are circumventing liability for damages that would otherwise apply
Did you know that mRNA COVID-19 vaccines aren’t vaccines in the medical and legal definition of a vaccine? They do not prevent you from getting the infection, nor do they prevent its spread. They’re really experimental gene therapies.
I discussed this troubling fact in a recent interview with molecular biologist Judy Mikovits, Ph.D. While the Moderna and Pfizer mRNA shots are labeled as “vaccines,” and news agencies and health policy leaders call them that, the actual patents for Pfizer’s and Moderna’s injections more truthfully describe them as “gene therapy,” not vaccines.
Definition of ‘Vaccine’
According to the U.S. Centers for Disease Control and Prevention,1 a vaccine is “a product that stimulates a person’s immune system to produce immunity to a specific disease, protecting the person from that disease.” Immunity, in turn, is defined as “Protection from an infectious disease,” meaning that “If you are immune to a disease, you can be exposed to it without becoming infected.”
Neither Moderna nor Pfizer claim this to be the case for their COVID-19 “vaccines.” In fact, in their clinical trials, they specify that they will not even test for immunity.
Unlike real vaccines, which use an antigen of the disease you’re trying to prevent, the COVID-19 injections contain synthetic RNA fragments encapsulated in a nanolipid carrier compound, the sole purpose of which is to lessen clinical symptoms associated with the S-1 spike protein, not the actual virus.
They do not actually impart immunity or inhibit transmissibility of the disease. In other words, they are not designed to keep you from getting sick with SARS-CoV-2; they only are supposed to lessen your infection symptoms if or when you do get infected.
As such, these products do not meet the legal or medical definition of a vaccine, and as noted by David Martin, Ph.D., in the video above, “The legal ramifications of this deception are immense.”
15 U.S. Code Section 41
As explained by Martin, 15 U.S. Code Section 41 of the Federal Trade Commission Act2 is the law that governs advertising of medical practices. This law, which dictates what you may and may not do in terms of promotion, has for many years been routinely used to shut down alternative health practitioners and companies.
“If this law can be used to shut down people of good will, who are trying to help others,” Martin says, “it certainly should be equally applied when we know deceptive medical practices are being done in the name of public health.”
Per this law, it is unlawful to advertise:
“… that a product or service can prevent, treat, or cure human disease unless you possess competent and reliable scientific evidence, including, when appropriate, well-controlled human clinical studies, substantiating that the claims are true at the time they are made.”3
What Constitutes ‘The Greater Good’?
Martin points to the 1905 Supreme Court ruling in Jacobson vs. Massachusetts,4 which essentially established that collective benefit supersedes individual benefit. To put it bluntly, it argued that it’s acceptable for individuals to be harmed by public health directives provided it benefits the collective.
Now, if vaccination is a public health measure that is supposed to protect and benefit the collective, then it would need to a) ensure that the individual who is vaccinated is rendered immune from the disease in question; and b) that the vaccine inhibits transmission of the disease.
Only if these two outcomes can be scientifically proven can you say that vaccination protects and benefits the collective — the population as a whole. This is where we run into problems with the mRNA “vaccines.”
Moderna’s SEC filings, which Martin claims to have carefully reviewed, specifies and stresses that its technology is a “gene therapy technology.” Originally, its technology was set up to be a cancer treatment, so more specifically, it’s a chemotherapy gene therapy technology.
As noted by Martin, who would raise their hand to receive prophylactic chemotherapy gene therapy for a cancer you do not have and may never be at risk for? In all likelihood, few would jump at such an offer, and for good reason.
Moreover, states and employers would not be able to mandate individuals to receive chemotherapy gene therapy for a cancer they do not have. It simply would not be legal. Yet, they’re proposing that all of humanity be forced to get gene therapy for COVID-19.
COVID-19 Vaccines — A Case of False Advertising
Now, if the COVID-19 vaccine really isn’t a vaccine, why are they calling it that? While the CDC provides a definition of “vaccine,” the CDC is not the actual law. It’s an agency empowered by the law, but it does not create law itself. Interestingly enough, it’s more difficult to find a legal definition of “vaccine,” but there have been a few cases. Martin provides the following examples:
• Iowa code — “Vaccine means a specially prepared antigen administered to a person for the purpose of providing immunity.” Again, the COVID-19 vaccines make no claim of providing immunity. They are only designed to lessen symptoms if and when you get infected.
• Washington state code — “Vaccine means a preparation of a killed or attenuated living microorganism, or fraction thereof …” Since Moderna and Pfizer are using synthetic RNA, they clearly do not meet this definition.
Being a manmade synthetic, the RNA used is not derived from anything that has at one point been alive, be it a whole microorganism or a fraction thereof. The statute continues to specify that a vaccine “upon immunization stimulates immunity that protects us against disease …”
So, in summary, “vaccine” and “immunity” are well-defined terms that do not match the end points specified in COVID-19 vaccine trials. The primary end point in these trials is: “Prevention of symptomatic COVID-19 disease.” Is that the same as “immunity”? No, it is not.
There Are More Problems Than One
But there’s another problem. Martin points out that “COVID-19 disease” has been defined as a series of clinical symptoms. Moreover, there’s no causal link between SARS-CoV-2, the virus, and the set of symptoms known as COVID-19.
How is that, you might ask? It’s simple, really. Since a vast majority of people who test positive for SARS-CoV-2 have no symptoms at all, they’ve not been able to establish a causal link between the virus and the clinical disease.
Here’s yet another problem: The primary end point in the COVID-19 vaccine trials is not an actual vaccine trial end point because, again, vaccine trial end points have to do with immunity and transmission reduction. Neither of those were measured.
What’s more, key secondary end points in Moderna’s trial include “Prevention of severe COVID-19 disease, and prevention of infection by SARS-CoV-2.” However, by its own admission, Moderna did not actually measure infection, stating that it was too “impractical” to do so.
That means there’s no evidence of this gene therapy having an impact on infection, for better or worse. And, if you have no evidence, you cannot fulfill the U.S. Code requirement that states you must have “competent and reliable scientific evidence … substantiating that the claims are true.”
Why Are They Calling Them Vaccines?
As noted by Martin, you cannot have a vaccine that does not meet a single definition of a vaccine. So, again, what would motivate these companies, U.S. health agencies and public health officials like Dr. Anthony Fauci to lie and claim that these gene therapies are in fact vaccines when, clearly, they are not?
If they actually called it what it is, namely “gene therapy chemotherapy,” most people would — wisely — refuse to take it. Perhaps that’s one reason for their false categorization as vaccines. But there may be other reasons as well.
Here, Martin strays into conjecture, as we have no proof of their intentions. He speculates that the reason they’re calling this experimental gene therapy technology a “vaccine” is because by doing so, they can circumvent liability for damages.
As long as the U.S. is under a state of emergency, things like PCR tests and COVID-19 “vaccines” are allowed under emergency use authorization. And as long as the emergency use authorization is in effect, the makers of these experimental gene therapies are not financially liable for any harm that comes from their use.
That is, provided they’re “vaccines.” If these injections are NOT vaccines, then the liability shield falls away, because there is no liability shield for a medical emergency countermeasure that is gene therapy.
So, by maintaining the illusion that COVID-19 is a state of emergency, when in reality it is not, government leaders are providing cover for these gene therapy companies so that they can get immunity from liability.
Under the Cover of ‘Emergency’
As noted by Martin, if state governors were to lift the state of emergency, all of a sudden the use of RT PCR testing would be in violation of 15 U.S. Code FTC Act, as PCR tests are not an approved diagnostic test.
“You cannot diagnose a thing [with something] that cannot diagnose a thing,” Martin says.“That a misrepresentation. That is a deceptive practice under the Federal Trade Commission Act. And they’re liable for deceptive practices.”
Importantly, there’s no waiver of liability under deceptive practices — even under a state of emergency. This would also apply to experimental gene therapies. The only way for these gene therapies to enjoy liability shielding is if they are vaccines developed in response to a public health emergency. There is no such thing as immunity from liability for gene therapies.
Propaganda and Vaccine Rollout Run by Same Company
Martin brings up yet another curious point. The middleman in Operation Warp Speed is a North Carolina defense contractor called ATI. It controls the rollout of the vaccine. But ATI also has another type of contract with the Department of Defense, namely managing propaganda and combating misinformation.
So, the same company in charge of manipulating the media to propagate government propaganda and censor counterviews is the same company in charge of the rollout of “vaccines” that are being unlawfully promoted.
“Listen,” Martin says. “This is a pretty straight-forward situation. You’re being lied to. Your own government is violating its own laws … They have thrown this book [15 U.S. Code Section 41] on more people than we can count.
They have shut down practitioners around the country, time and time again, for violating what are called ‘deceptive practices in medical claims’ … Guess what? They’re doing exactly that thing.”
Martin urges listeners to forward his video to your state attorney, governor, representatives and anyone else that might be in a position to take affirmative action to address and correct this fraud.
Defense contractors are violating FTC law, and gene therapy companies — not vaccine manufacturers — are conducting experimental trials under deceptive medical practices. They’re making claims of being “vaccines” without clinical proof, and must be held accountable for their deceptive marketing and medical practices.
CDC Owns Coronavirus Patents
On a side note, the CDC appears to be neck-deep in this scam pandemic, and is therefore wholly unsuitable to investigate the side effects of these experimental COVID-19 therapies. As noted by Martin, it’s like having a bank robber investigate its own crime.
Details about this came out in the documentary “Plandemic,” in which Martin explained how the CDC has broken the law — in one way or another — related to its patenting of the 2003 SARS virus.
Martin is a national intelligence analyst and founder of IQ100 Index, which developed linguistic genomics, a platform capable of determining the intent of communications. In 1999, IBM digitized 1 million U.S. patents, which allowed Martin’s company to conduct a review of all these patents, sending him down a proverbial “rabbit trail” of corruption.
In 2003, Asia experienced an outbreak of SARS. Almost immediately, scientists began racing to patent the virus. Ultimately, the CDC nabbed ownership of SARS-CoV (the virus responsible for SARS) isolated from humans.
So, the CDC actually owns the entire genetic content of that SARS virus. It’s patented under U.S. patent 7776521. They also own patents for detection methods, and for a kit to measure the virus.
U.S. patent 7279327,5 filed by the University of North Carolina at Chapel Hill, describes methods for producing recombinant coronaviruses. Ralph Baric, Ph.D., a professor of microbiology and immunology who is famous for his chimeric coronavirus research, is listed as one of the three inventors, along with Kristopher Curtis and Boyd Yount.
According to Martin, Fauci, Baric and the CDC “are at the hub” of the whole COVID-19 story. “In 2002, coronaviruses were recognized as an exploitable mechanism for both good and ill,” Martin says, and “Between 2003 and 2017, they [Fauci, Baric and CDC] controlled 100% of the cash flow to build the empire around the industrial complex of coronavirus.”
How the CDC Broke the Law
The key take-home message Martin delivers in “Plandemic” is that there’s a distinct problem with the CDC’s patent on SARS-CoV isolated from humans, because, by law, naturally occurring DNA segments are prohibited from being patented.
The law clearly states that such segments are “not patent eligible merely because it has been isolated.” So, either SARS-CoV was manmade, which would render the patent legal, or it’s natural, thus rendering the patent on it illegal.
However, if the virus was manufactured, then it was created in violation of biological weapons treaties and laws. This includes the Biological Weapons Anti-Terrorism Act of 1989, passed unanimously by both houses of Congress and signed into law by George Bush Sr., which states:6
“Whoever knowingly develops, produces, stockpiles, transfers, acquires, retains, or possesses any biological agent, toxin, or delivery system for use as a weapon, or knowingly assists a foreign state or any organization to do so, shall be fined under this title or imprisoned for life or any term of years, or both. There is extraterritorial Federal jurisdiction over an offense under this section committed by or against a national of the United States.”
So, as noted by Martin in the documentary, regardless of which scenario turns out to be true, the CDC has broken the law one way or another, either by violating biological weapons laws, or by filing an illegal patent. Even more egregious, May 14, 2007, the CDC filed a petition with the patent office to keep their coronavirus patent confidential.
Now, because the CDC owns the patent on SARS-CoV, it has control over who has the ability to make inquiries into the coronavirus. Unless authorized, you cannot look at the virus, you cannot measure it or make tests for it, since they own the entire genome and all the rest.
“By obtaining the patents that restrained anyone from using it, they had the means, the motive, and most of all, they had the monetary gain from turning coronavirus from a pathogen to a profit,” Martin says.
Dangers of mRNA Gene Therapy
I’ve written many articles detailing the potential and expected side effects of these gene therapy “vaccines.” If all of this is new to you, consider reviewing “How COVID-19 Vaccine Can Destroy Your Immune System,” “Seniors Dying After COVID Vaccine Labeled as Natural Causes” and “Side Effects and Data Gaps Raise Questions on COVID Vaccine.”
In the lecture above, Dr. Simone Gold — founder of America’s Frontline Doctors, which has been trying to counter the false narrative surrounding hydroxychloroquine — reviews the dangers discovered during previous coronavirus vaccine trials, and the hazards of current mRNA gene therapies, including antibody-dependent immune enhancement.
Antibody-dependent immune enhancement results in more severe disease when you’re exposed to the wild virus, and increases your risk of death. The synthetic RNA and the nanolipid its encased in may also have other, more direct side effects. As explained by Mikovits in our recent interview:
“Normally, messenger RNA is not free in your body because it’s a danger signal. The central dogma of molecular biology is that our genetic code, DNA, is transcribed, written, into the messenger RNA. That messenger RNA is translated into protein, or used in a regulatory capacity … to regulate gene expression in cells.
So, taking a synthetic messenger RNA and making it thermostable — making it not break down — [is problematic]. We have lots of enzymes (RNAses and DNAses) that degrade free RNA and DNA because, again, those are danger signals to your immune system. They literally drive inflammatory diseases.
Now you’ve got PEG, PEGylated and polyethylene glycol, and a lipid nanoparticle that will allow it to enter every cell of the body and change the regulation of our own genes with this synthetic RNA, part of which actually is the message for the gene syncytin …
Syncytin is the endogenous gammaretrovirus envelope that’s encoded in the human genome … We know that if syncytin … is expressed aberrantly in the body, for instance in the brain, which these lipid nanoparticles will go into, then you’ve got multiple sclerosis.
The expression of that gene alone enrages microglia — literally inflames and dysregulates the communication between the brain microglia — which are critical for clearing toxins and pathogens in the brain and the communication with astrocytes.
It dysregulates not only the immune system, but also the endocannabinoid system, which is the dimmer switch on inflammation. We’ve already seen multiple sclerosis as an adverse event in the clinical trials … We also see myalgic encephalomyelitis. Inflammation of the brain and the spinal cord …”
Making matters worse, the synthetic mRNA also has an HIV envelope expressed in it, which can cause immune dysregulation. As we discussed in previous interviews, SARS-CoV-2 has been engineered in the lab with gain-of-function research that included introducing the HIV envelope into the spike protein.
Are You in a High-Risk Group for Side Effects?
Mikovits’ hypothesis is that those who are most susceptible to severe neurological side effects and death from the COVID-19 vaccines are those who have previously been injected with XMRVs, borrelia, babesia or mycoplasma through contaminated vaccines, resulting in chronic disease, as well as anyone with an inflammatory disease like rheumatoid arthritis, Parkinson’s disease or chronic Lyme disease, for example, and anyone with an acquired immune deficiency from any pathogens and environmental toxins.
The chart below lists 35 diseases that are likely to render you more susceptible to severe side effects or death from COVID-19 gene therapy injections.
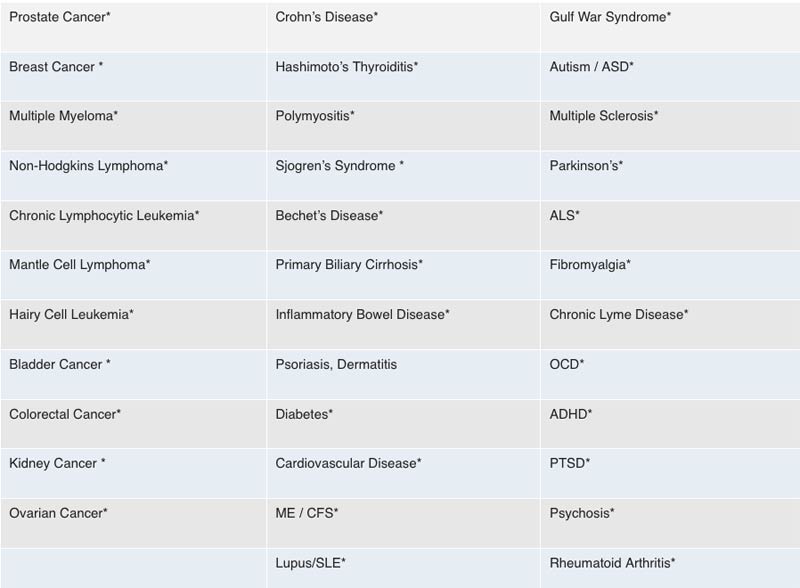
Many of the symptoms now being reported are suggestive of neurological damage. They have severe dyskinesia (impairment of voluntary movement), ataxia (lack of muscle control) and intermittent or chronic seizures. Many cases detailed in personal videos on social media are quite shocking. According to Mikovits, these side effects are due to neuroinflammation, a dysregulated innate immune response, and/or a disrupted endocannabinoid system.
Another common side effect from the vaccine we’re seeing is allergic reactions, including anaphylactic shock. A likely culprit in this is PEG (polyethylene glycol), which an estimated 70% of Americans are allergic to.
Experimental Gene Therapy Is a Bad Idea
Circling back to where we began, COVID-19 vaccines are not vaccines. They are experimental gene therapies that are falsely marketed as vaccines, likely to circumvent liability. World governments and global and national health organizations are all complicit in this illegal deception and must be held accountable.
Ask yourself the question Martin asked in his video: Would you agree to take an experimental chemotherapy gene therapy for a cancer you do not have? If the answer is no, then why would you even consider lining up for an experimental gene therapy for COVID-19 — a set of clinical symptoms that haven’t even been causally linked to SARS-CoV-2?
These injections are not vaccines. They do not prevent infection, they do not render you immune, and they do not prevent transmission of the disease. Instead, they alter your genetic coding, turning you into a viral protein factory that has no off-switch. What’s happening here is a medical fraud of unprecedented magnitude, and it really needs to be stopped before it’s too late for a majority of people.
_______________________
**Comment**
Remember, the same people/organizations behind the Lyme/MSIDS fraud are behind the current fraud surrounding COVID. They’ve been committing fraud for over 40 years and they haven’t stopped.
PLEASE NOTE CHRONIC LYME IS LISTED AS HIGH RISK FOR SIDE EFFECTS.
The entire house of cards is built upon faulty testing. Please see the following quote from the creator of the PCR test who states this fact:

For an eye-opening video, please see: https://www.bitchute.com/video/KO495u7J749A/ Title:
You’ll Never Guess What’s in the Tests
- It discusses the nanotechnology used in the COVID testing swab: https://www.sciencedirect.com/science/article/abs/pii/S0169409X08002640 (Are we being tested or vaccinated and nano chipped without our knowledge?) A Morgellons patient states that the same silver fibers coming out of her body are found in the COVID test swab. https://www.morgellonsexposed.com A doctor discusses this at about 12:00 (these fibers won’t burn).
- Gene editing
- At about 17:50 Dr. Lorraine Day states that this genetic modification makes you patentable. She states that the COVID test vaccinates you & hooks you up to the Cloud. The following patents are revealing: https://patents.google.com/patent/US10163055B2/en Abstract: Methods, systems, and products provide interfaces between intrahost networks and interhost networks within biological hosts. Neuroregional translations are performed to route communications to and from the biological hosts. Bioregional translations may also be performed to route communications to and from the biological hosts. Also see: https://madisonarealymesupportgroup.com/2020/04/29/gates-patent-for-body-activity-data-apparatus/ Abstract: “Human body activity associated with a task provided to a user(1) may be used in a mining process of a cryptocurrency system. A server may provide a task to a device of a user which is communicatively coupled to the server(2). A sensor communicatively coupled to or comprised in the device(3) of the user may sense body activity of the user. Body activity data may be generated based on the sensed body activity of the user. The cryptocurrency system communicatively coupled to the device of the user may verify if the body activity data satisfies one or more conditions set by the cryptocurrency system(4), and award cryptocurrency to the user whose body activity data is verified.”(5)
- The video states a clue to the anaphylaxis being seen is explained by the following: https://www.sciencedaily.com/releases/2018/04/180404114727.htm Nanoparticles may cause DNA damage to brain cells across a cellular barrier
- Around 29:00 Dr. Martin goes into how the COVID injection is NOT a vaccine but a pathogen creator, and that the use of the term “vaccine” for this is unconscionable. A “packet of technology” is being inserted into the human body.
- Regarding AIDS and whistleblower Dr. Strecker: https://www.labsavvy.org/the-strecker-memorandum.html The AIDS debacle along with many other virus ‘pandemics’ are discussed in the book Virus Mania: How the Medical Industry Continually Invents Epidemics, making Billion-dollar Profits at Our Expense. For a brief, old interview with Kary Mullis, PCR creator: https://beforeitsnews.com/agenda-21/2020/12/kary-mullis-inventor-of-pcr-technique-about-science-fauci-and-their-agenda-engger-subtitles- Mullis is obviously not a fan of Dr. Fauci and he never believed the story on AIDS. For a lengthier article: https://madisonarealymesupportgroup.com/2020/05/07/was-the-covid-19-test-meant-to-detect-a-virus/
- The Chinese biological lab in Wuhan is owned by GlaxoSmith Kline which merged with and owns 70% controlling stake in Pfizer (a COVID injection manufacturer). Pfizer manages the finances of Black Rock & Black Rock owns stakes in both Pfizer, AstraZeneca, and another company developing a COVID injection. Black Rock also has a partnership with Thompson Reuters, a multimillion dollar national media conglomerate, which essentially controls all the news.
- The COVID-19 test kit (902780) was ordered back in 2018 by many countries including the U.S.
- The fact people injected with the COVID shot are testing positive for HIV is not surprising when you understand the shot’s ability to cause autoimmunity. Dr. Shiva Ayyadurai states Fauci is behind the big lie that HIV causes AIDS: https://madisonarealymesupportgroup.com/2020/04/23/dr-shiva-ayyadurai-mit-phd-on-covid-19-the-truth-leads-to-a-solution-that-has-nothing-to-do-with-vaccines-and-shutting-down-the-country/ Fauci has ruled NIAID for 7 presidencies & doles out all the research dollars.
- The sterility issue is also addressed, as is here: https://madisonarealymesupportgroup.com/2020/08/24/gsk-whistleblower-covid-vaccine-caused-sterility-in-97-of-women/
- There’s an important section on anaphylaxis and a bit of history on Charles Richet’s work found here: https://www.nobelprize.org/prizes/medicine/1913/richet/lecture/ In short, it’s all about the importance of the interval between injections. Experiments by 1902 showed: (1) a subject that had a previous injection is far more sensitive than a new subject; (2) that the symptoms characteristic of the second injection, namely swift and total depression of the nervous system, do not in any way resemble the symptoms characterizing the first injection; (3) a three or four week period must elapse before the anaphylactic state results. This is the period of incubation. This 3 weeks is now what the UK is recommending for the second dose.
- The video goes through a plethora of deaths after the shot. This video was shown as well, which I posted before: https://madisonarealymesupportgroup.com/2021/01/28/cna-seniors-are-dying-like-flies-after-covid-injections/ Many are being told not to report injury and death to VAERS. Serious injuries are given as well, and here is an ongoing list: https://madisonarealymesupportgroup.com/2020/12/21/warning-3150-injuries-in-1st-week-of-covid-vaccines-among-american-healthcare-workers-pregnant-women-included/
- This alleged virus has never been isolated, has the same survival rate as the flu, many are asymptomatic or have mild symptoms, and those that die are typically elderly with comorbidities. Also – the seasonal flu has completely disappeared: https://madisonarealymesupportgroup.com/2020/11/03/why-is-cdc-scaring-us-to-death/ Yet our public ‘authorities’ want us to believe that we must get this shot due to some horrific threat.
- At about 1:43 Dr. Cowan describes 31 papers from peer-reviewed journals claiming isolation of COVID-19. Here’s another similar article: https://blog.nomorefakenews.com/2020/10/19/dr-tom-cowan-explores-the-covid-virus-invented-out-of-sheer-nonsense/ He states there is NOT ONE study proving COVID-19 exists. His explanation needs to be understood and shared. This is the root of the issue scientifically. Please listen to his explanation as he does a fantastic job explaining this to the lay-man.
COVID-19, similarly to HIV and other viruses, has never been “separated from everything else,” and is a laboratory artifact, a set of lab-tortured antigens around which a “test” was built: https://madisonarealymesupportgroup.com/2020/05/07/was-the-covid-19-test-meant-to-detect-a-virus/


