Do you have all the symptoms of Lyme disease but test negative? Besides the fact the standard test for Lyme misses half the cases, there may be another reason—Borrelia miyamotoi.
A new study out of San Francisco State University (SFSU) has found more Borrelia miyamotoi than Borrelia burgdorferi (Lyme disease) in Ixodes pacificus ticks in some areas of Northern California.
Borrelia miyamotoi disease poses a difficult problem for patients and doctors who are unfamiliar with it. Its symptoms may present as viral-like or Lyme-like and there is currently no FDA-approved test.
What is Borrelia miyamotoi?
The family of tick-borne Borrelia is divided into two broad groups that correspond to the disease manifestations they cause in humans: Lyme disease or relapsing fever. Borrelia miyamotoi is in the tick-borne relapsing fever group (TBRF).
Unlike B. miyamotoi, most North American TBRF species (B. hermsii, B. turicatae, B. parkeri), are transmitted by soft-bodied ticks.
B. miyamotoi is different. It’s transmitted by the same blacklegged tick that carries Lyme disease and several other pathogens.
Furthermore, B. miyamotoi can be transmitted from the adult female tick to her eggs. This is called transovarial transmission. It means that baby ticks can be infected with B. miyamotoi from the get-go.
In contrast, blacklegged ticks don’t start out being infected with Lyme. They must acquire the infection by feeding on an animal that carries it—such as a mouse or squirrel.
The research and findings
For the SFSU study, researchers collected 1,358 Ixodes pacificus ticks and 370 rodents from eight different sites in Northern California from April to May 2018. The ticks included 894 larvae, 281 nymphs and 183 adults.
On average the following tested positive for B. miyamotoi:
- 0.11% of the larvae
- 4.98% of nymphs
- 8.74% of the adult ticks
With the prevalence of infection increasing at each life stage of the tick, and each stage including one blood meal, the researchers estimate that horizontal transmission (from mammal to tick) accounts for 97% of B. miyamotoi infection in nymphal ticks and 43% of infection in adult ticks.
Interestingly, the researchers detected B. burgdorferi in 9.86% of nymph and 2.33% of the adult I. pacificus ticks—a decrease in prevalence of 76% from one life stage to the next. In addition, an average of 5.21% of the mice had blood that tested positive for B. miyamotoi.
During the same time, researchers also took skin biopsies from the ears of the rodents they had collected, and examined them for the presence of miyamotoi infection and Lyme. They found more B. miyamotoi in the blood samples and more B. burgdorferi in the skin.
This matches with other types of TBRF, which are more easily detected in human blood samples, especially when the person is symptomatic. In contrast, Lyme disease is harder to detect in blood samples after it disseminates throughout the body.
The SFSU authors conclude, “the human risk of B. miyamotoi in the western USA may be even greater than B. burgdorferi in certain disease hot spots.”
How this study compares
The SFSU study only included counties around the San Francisco Bay Area. However, a prior California Department of Public Health 12-year study found B. miyamotoi in I. pacificus ticks in 24 out of 48 California counties.
Another study published in 2018 by Professor Robert Lane, along with researchers at UC Berkeley and Yale, examined a biobank of frozen blood samples. The results showed that 26 out of 101 samples collected from residents of Mendocino County, California, in the 1980s were reactive for relapsing fever borreliosis, including B. miyamotoi.
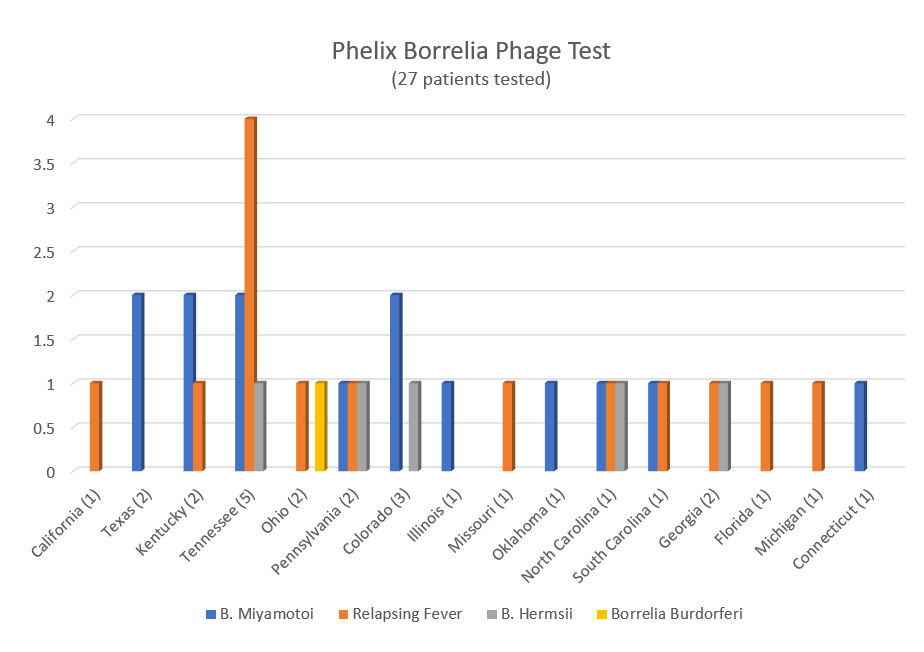
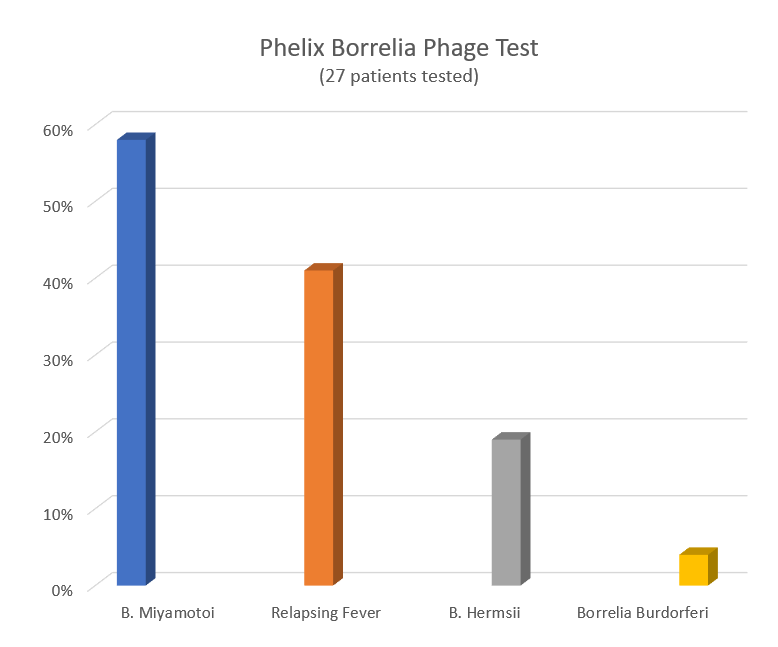
Similarly, another study published in 2018 looked at blood samples drawn from patients in 25 California counties. The results showed positive testing for TBRF in 16 of those counties, with the highest rates of positivity coming from Santa Clara, Alameda, Marin and San Francisco.
That study used a novel laboratory technique that can detect multiple species of TBRF. Co-author Jyotsna Shaw, of IGeneX, Inc., says “we hope that these techniques can be used to develop accurate FDA-approved tests for the future.”
A hidden epidemic?
B. miyamotoi was discovered in ticks more than 20 years ago in Japan. But it wasn’t proven to cause illness in humans until 2011, when the first human cases were reported in Russia.
It is now known to be widespread in North America, wherever Ixodes scapularis ticks and Ixodes pacificus ticks are found.
Unfortunately, Borrelia miyamotoi disease (BMD) is not reportable to the CDC, and the standard test for Lyme disease will not detect it. Because of this, many patients are likely going undiagnosed, and therefore we really have no idea how widespread the illness is.
The symptoms of Borrelia Miyamotoi Disease (BMD)
(according to the CDC)
Symptoms
- Fever
- Chills
- Fatigue
- Severe headache
- Muscle or joint pain
- Dizziness, confusion, vertigo (uncommon)
- Rash (uncommon)
- Shortness of breath (uncommon)
- Nausea, abdominal pain, diarrhea, and anorexia (uncommon)
A recent feature in the New York Times discusses a difficult case where a patient tested negative for Lyme and many other conditions. Medical detective work by Dr. Brian Fallon of the Columbia Lyme and Tick-Borne Disease Center eventually determined that she suffered from B. miyamotoi. (She tested negative for Lyme disease. So what was wrong?)
LymeSci is written by Lonnie Marcum, a Licensed Physical Therapist and mother of a daughter with Lyme. Follow her on Twitter: Email her at: lmarcum@lymedisease.org .
References
Sambado, S., J. Salomon, A. Crews, and A. Swei. (2020) Mixed transmission modes promote persistence of an emerging tick-borne pathogen. Ecosphere 11(6):e03171. 10.1002/ecs2.3171
Krause PJ, Carroll M, Fedorova N, Brancato J, Dumouchel C, Akosa F, et al. (2018) Human Borrelia miyamotoi infection in California: Serodiagnosis is complicated by multiple endemic Borrelia species. PLoS ONE 13(2): e0191725. https://doi.org/10.1371/journal.pone.0191725
Padgett K, Bonilla D, Kjemtrup A, Vilcins I-M, Yoshimizu MH, Hui L, et al. (2014) Large Scale Spatial Risk and Comparative Prevalence of Borrelia miyamotoi and Borrelia burgdorferi Sensu Lato in Ixodes pacificus. PLoS ONE 9(10): e110853. https://doi.org/10.1371/journal.pone.0110853
Jeomhee Mun, Rebecca J. Eisen, Lars Eisen, Robert S. Lane (2006) Detection of a Borrelia miyamotoi Sensu Lato Relapsing-Fever Group Spirochete from Ixodes pacificus in California , Journal of Medical Entomology, 43 (1) 120–123, https://doi.org/10.1093/jmedent/43.1.120
Geoffrey E Lynn, Christine B Graham, Kalanthe Horiuchi, Lars Eisen, Tammi L Johnson, Robert S Lane, Rebecca J Eisen (2018) Prevalence and Geographic Distribution of Borrelia miyamotoi in Host-Seeking Ixodes pacificus (Acari: Ixodidae) Nymphs in Mendocino County, California, Journal of Medical Entomology, 55(3), 711–716, https://doi.org/10.1093/jme/tjx258
Borrelia miyamotoi Disease | Tick-borne Diseases | Ticks | CDC https://www.cdc.gov/ticks/tickbornediseases/borrelia-miyamotoi.html
Test Order | Submitting Specimens to CDC | Infectious Diseases Laboratories | CDC https://www.cdc.gov/laboratory/specimen-submission/detail.html?CDCTestCode=CDC-10532 via @CDCgov
__________________
**Comment**




 1,2
1,2